QUESTION IMAGE
Question
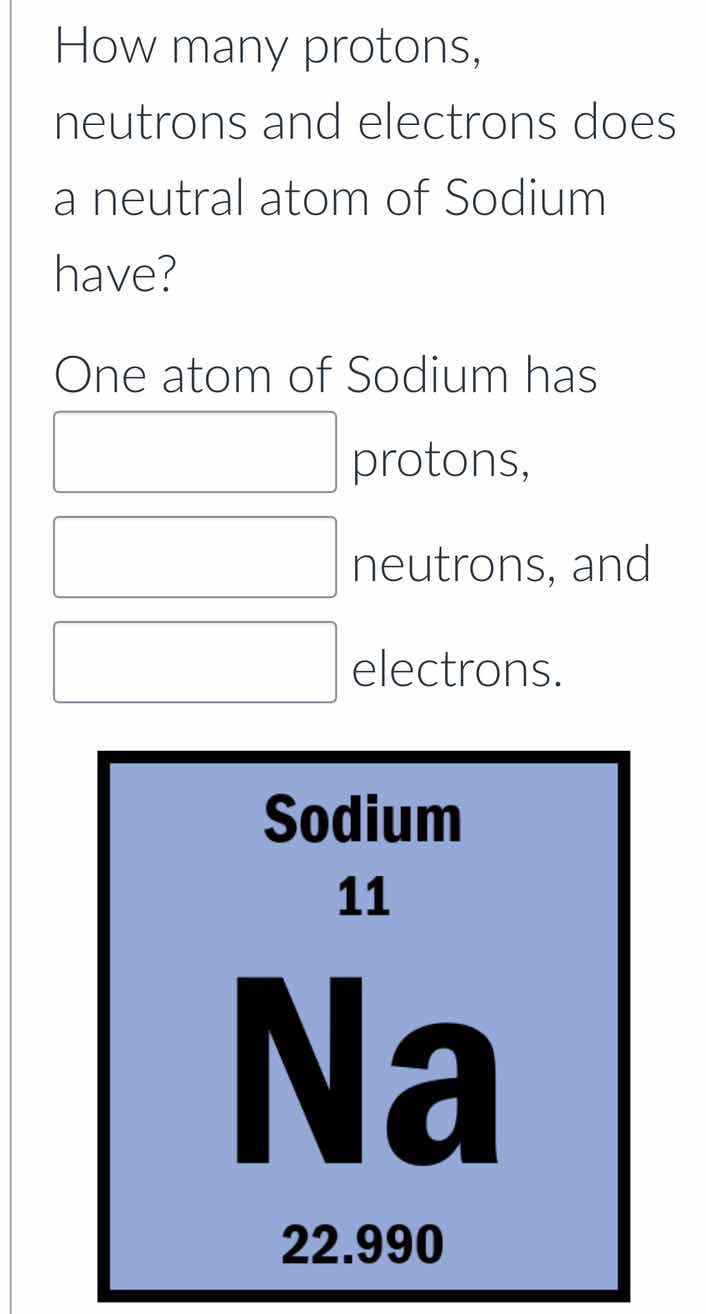
how many protons, neutrons and electrons does a neutral atom of sodium have? one atom of sodium has blank protons, blank neutrons, and blank electrons. image of sodiums periodic table entry: sodium, 11, na, 22.990
Step1: Find proton count
The atomic number (11) equals proton count: $11$
Step2: Calculate neutron count
Round atomic mass to 23, subtract protons: $23 - 11 = 12$
Step3: Find electron count
Neutral atom has equal electrons/protons: $11$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
One atom of Sodium has 11 protons, 12 neutrons, and 11 electrons.