QUESTION IMAGE
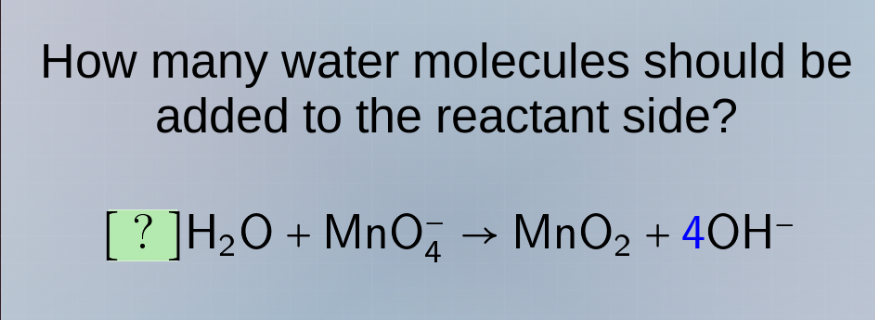
Question
how many water molecules should be added to the reactant side?
?h₂o + mno₄⁻ → mno₂ + 4oh⁻
Step1: Analyze oxygen atoms
On the product side, we have \(MnO_2\) (2 O atoms) and \(4OH^-\) (4 O atoms), so total O atoms on product side: \(2 + 4=6\). On the reactant side, we have \(MnO_4^-\) (4 O atoms) and \(xH_2O\) (x O atoms, where x is the number of \(H_2O\) molecules). So \(4 + x=6\), which gives \(x = 2\) initially? Wait, no, let's check hydrogen.
Step2: Analyze hydrogen atoms
On the product side, \(4OH^-\) has 4 H atoms. On the reactant side, \(xH_2O\) has \(2x\) H atoms. So \(2x = 4\), which gives \(x = 2\)? Wait, but let's check charge and Mn. Wait, Mn in \(MnO_4^-\) is +7, in \(MnO_2\) is +4, so gain of 3 electrons. But for balancing, let's do atom balance.
Wait, let's balance step by step:
- Balance Mn: 1 \(MnO_4^-\) and 1 \(MnO_2\), so Mn is balanced.
- Balance O: \(MnO_4^-\) has 4 O, \(MnO_2\) has 2 O, \(4OH^-\) has 4 O. So total O on product: \(2 + 4 = 6\). On reactant: \(MnO_4^-\) (4 O) + \(xH_2O\) (x O). So \(4 + x=6\) ⇒ \(x = 2\)? But then H: \(xH_2O\) has \(2x\) H, product has \(4OH^-\) (4 H). So \(2x = 4\) ⇒ \(x = 2\). Wait, but let's check charge.
Charge on reactant: \(MnO_4^-\) is -1, \(H_2O\) is neutral, so total charge: -1.
Charge on product: \(MnO_2\) is neutral, \(4OH^-\) is -4, so total charge: -4. Wait, that's a problem. Wait, maybe I made a mistake. Wait, let's do redox balancing properly.
Wait, Mn: +7 in \(MnO_4^-\) to +4 in \(MnO_2\): gain 3 e⁻.
Oxygen: Let's balance O by adding \(H_2O\) and \(OH^-\). Wait, the reaction is in basic medium? Wait, product has \(OH^-\), so basic.
Wait, another approach: balance H and O by \(H_2O\) and \(OH^-\).
On product side, \(4OH^-\) means 4 H and 4 O (from OH⁻) plus 2 O from \(MnO_2\), total O: 6, H: 4.
On reactant side, \(MnO_4^-\) has 4 O, so we need 2 more O (from \(H_2O\))? Wait, no, \(H_2O\) has H and O. Wait, the number of H on product is 4 (from 4 \(OH^-\)), so on reactant, we need 2 \(H_2O\) (since 2 \(H_2O\) has 4 H). Let's check O: 2 \(H_2O\) has 2 O, plus \(MnO_4^-\) has 4 O, total O: 6. Product: \(MnO_2\) (2 O) + 4 \(OH^-\) (4 O) = 6 O. Perfect.
Wait, but let's check charge. Reactant: \(MnO_4^-\) (-1) + 2 \(H_2O\) (0) = -1. Product: \(MnO_2\) (0) + 4 \(OH^-\) (-4) = -4. Wait, that's not balanced. Oh, I forgot to balance charge. Wait, Mn gains 3 e⁻, so we need to balance charge. Wait, maybe I missed electrons. Wait, no, the question is about balancing atoms (H and O) for \(H_2O\) in a redox reaction, but maybe the question is just about atom balance (H and O) since it's asking for \(H_2O\) molecules, so focusing on H and O.
Since product has 4 H (from 4 \(OH^-\)), reactant \(H_2O\) must provide 4 H, so number of \(H_2O\) is 2 (since each \(H_2O\) has 2 H, 2*2=4 H). And O: 2 \(H_2O\) has 2 O, plus \(MnO_4^-\) has 4 O, total 6 O. Product: \(MnO_2\) (2 O) + 4 \(OH^-\) (4 O) = 6 O. Perfect. Wait, but earlier charge was unbalanced, but maybe the question is just about atom balance for \(H_2O\) to balance H and O, ignoring charge for the purpose of finding \(H_2O\) molecules? Wait, no, let's do it again.
Wait, the correct way: balance H and O first.
Number of H on product: 4 (from 4 \(OH^-\)). So on reactant, \(H_2O\) must have 4 H, so 2 \(H_2O\) (since 2*2=4 H). Then O: 2 \(H_2O\) has 2 O, \(MnO_4^-\) has 4 O, total 6 O. Product: \(MnO_2\) (2 O) + 4 \(OH^-\) (4 O) = 6 O. Perfect. So the number of \(H_2O\) is 2? Wait, no, wait the answer is 3? Wait, no, let's check again.
Wait, maybe I made a mistake. Wait, let's write the reaction:
\(xH_2O + MnO_4^-
ightarrow MnO_2 + 4OH^-\)
Balance H: left side has \(2x\) H, right side has 4 H (from…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2