QUESTION IMAGE
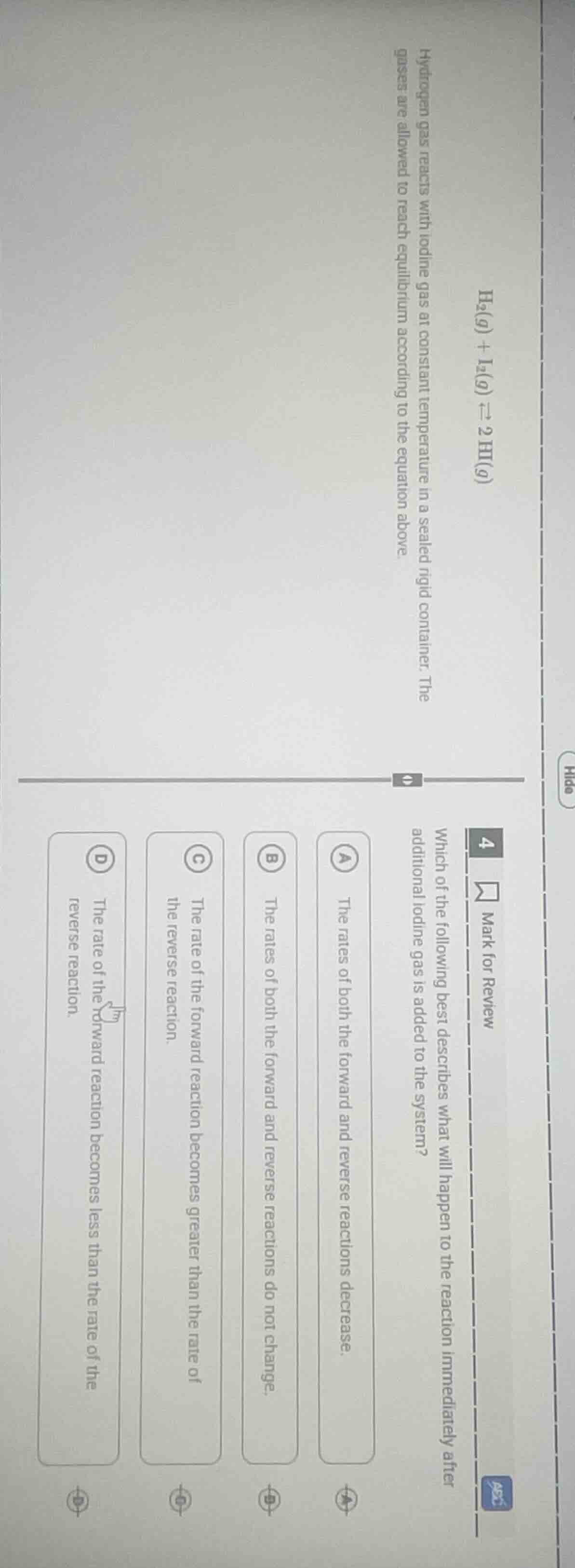
Question
hydrogen gas reacts with iodine gas at constant temperature in a sealed rigid container. the gases are allowed to reach equilibrium according to the equation above.
- which of the following best describes what will happen to the reaction immediately after additional iodine gas is added to the system?
a. the rates of both the forward and reverse reactions decrease.
b. the rates of both the forward and reverse reactions do not change.
c. the rate of the forward reaction becomes greater than the rate of the reverse reaction.
d. the rate of the forward reaction becomes less than the rate of the reverse reaction.
To solve this, we use Le Chatelier's principle. The reaction is \( \ce{H_{2}(g) + I_{2}(g)
ightleftharpoons 2HI(g)} \). When additional \( \ce{I_{2}(g)} \) (a reactant) is added, the system will try to counteract this change by favoring the forward reaction (to consume the added \( \ce{I_{2}} \)).
- Option A: Rates don't decrease; adding a reactant increases the forward rate initially, so A is wrong.
- Option B: Adding a reactant changes the rates (forward rate increases), so B is wrong.
- Option C: When \( \ce{I_{2}} \) is added, the forward reaction rate increases (to use up the extra \( \ce{I_{2}} \)). At this instant, the reverse reaction rate is still at its previous equilibrium value (before \( \ce{I_{2}} \) was added), so the forward rate is greater than the reverse rate. This matches.
- Option D: The forward rate should be greater (not less) than the reverse rate immediately after adding \( \ce{I_{2}} \), so D is wrong.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. The rate of the forward reaction becomes greater than the rate of the reverse reaction.