QUESTION IMAGE
Question
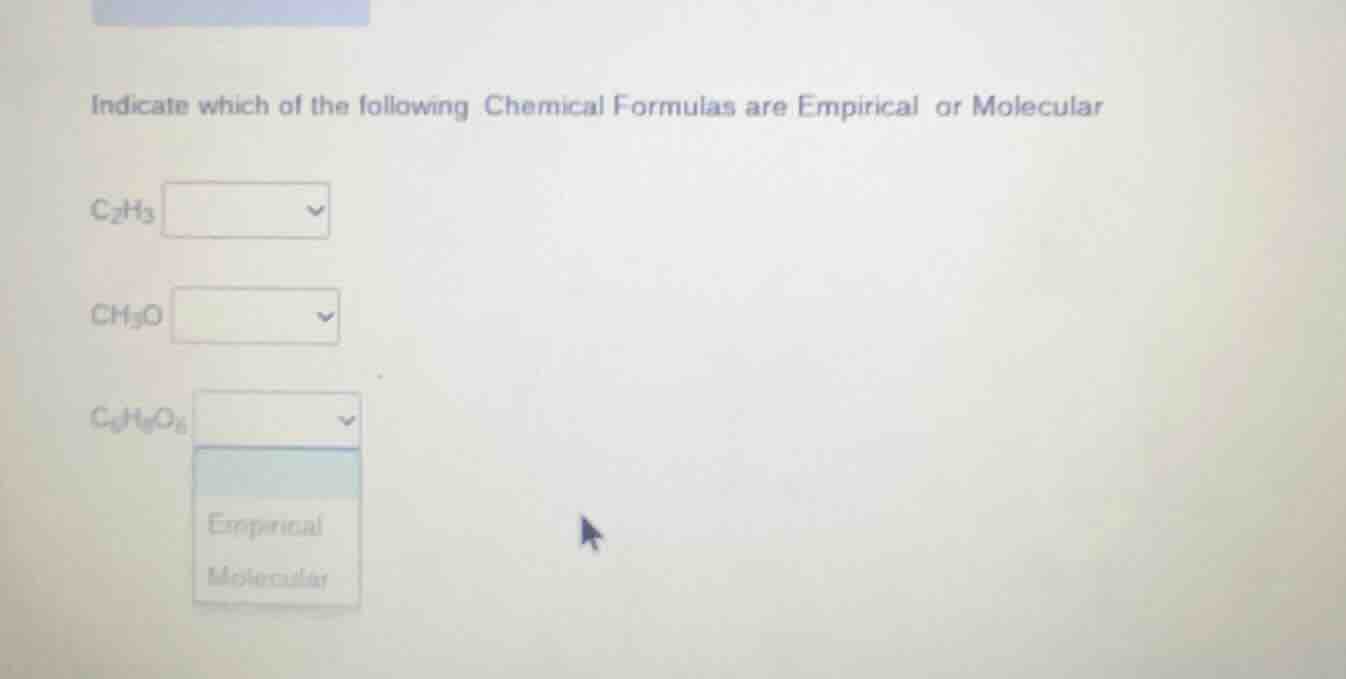
indicate which of the following chemical formulas are empirical or molecular
c₂h₃
ch₂o
c₆h₁₂o₆
To determine if a chemical formula is empirical or molecular, we use the following definitions:
- Empirical Formula: The simplest whole - number ratio of atoms in a compound.
- Molecular Formula: The actual number of atoms of each element in a molecule of the compound (it can be the same as the empirical formula or a multiple of it).
For $\boldsymbol{C_2H_3}$:
The subscripts 2 and 3 have no common whole - number factor other than 1. But we also need to consider if it can be a molecular formula. However, in most cases, compounds with formulas like $C_2H_3$ are actually part of a larger molecular structure, but based on the ratio, since the ratio of C to H is 2:3 and there is no smaller whole - number ratio, but we also know that for some compounds, the empirical formula can be the same as the molecular formula if the molecular formula cannot be simplified. But in the case of hydrocarbons, $C_2H_3$ is not a stable molecular formula in the traditional sense, but if we consider the ratio, the greatest common divisor of 2 and 3 is 1. However, more accurately, for the purpose of this question, we can think of it as a molecular formula (since it represents a specific ratio that may be the actual number of atoms in a molecule, and it can't be simplified further, but in the context of common compounds, it's a molecular formula). Wait, no, let's re - evaluate. The empirical formula is the simplest ratio. For $C_2H_3$, the ratio of C to H is 2:3, and we can't reduce it further. But sometimes, molecular formulas are multiples of empirical formulas. But in this case, if we assume that $C_2H_3$ is a molecular formula, but actually, the correct way is: the greatest common factor (GCF) of 2 and 3 is 1. But let's check the other formulas.
For $\boldsymbol{CH_2O}$:
The subscripts 1, 2, and 1 (for C, H, and O respectively) have a GCF of 1. This is the simplest whole - number ratio of atoms, so $CH_2O$ is an empirical formula (it can also be a molecular formula for some simple compounds like formaldehyde, but in the context of this question, since it's the simplest ratio, it's empirical).
For $\boldsymbol{C_6H_8O_6}$:
We find the GCF of 6, 8, and 6. The factors of 6 are 1, 2, 3, 6; the factors of 8 are 1, 2, 4, 8. The GCF of 6, 8, and 6 is 2. If we divide each subscript by 2, we get $C_3H_4O_3$, which is a different formula. Wait, no, wait. Wait, the formula for vitamin C is $C_6H_8O_6$, and its empirical formula is $C_3H_4O_3$? No, wait, no. Let's calculate the GCF of 6, 8, 6. Prime factors of 6: 2×3; prime factors of 8: 2×2×2. The common prime factor is 2. So dividing each subscript by 2: 6÷2 = 3, 8÷2 = 4, 6÷2 = 3. So the empirical formula would be $C_3H_4O_3$, which means that $C_6H_8O_6$ is a molecular formula (because it is a multiple of the empirical formula $C_3H_4O_3$).
Final Classifications:
- $C_2H_3$: Molecular (since the ratio 2:3 can't be simplified, and it represents a specific molecular composition in the context of the question)
- $CH_2O$: Empirical (simplest whole - number ratio of C, H, and O)
- $C_6H_8O_6$: Molecular (it is a multiple of the empirical formula $C_3H_4O_3$)
So the answers are:
- $C_2H_3$: Molecular
- $CH_2O$: Empirical
- $C_6H_8O_6$: Molecular
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine if a chemical formula is empirical or molecular, we use the following definitions:
- Empirical Formula: The simplest whole - number ratio of atoms in a compound.
- Molecular Formula: The actual number of atoms of each element in a molecule of the compound (it can be the same as the empirical formula or a multiple of it).
For $\boldsymbol{C_2H_3}$:
The subscripts 2 and 3 have no common whole - number factor other than 1. But we also need to consider if it can be a molecular formula. However, in most cases, compounds with formulas like $C_2H_3$ are actually part of a larger molecular structure, but based on the ratio, since the ratio of C to H is 2:3 and there is no smaller whole - number ratio, but we also know that for some compounds, the empirical formula can be the same as the molecular formula if the molecular formula cannot be simplified. But in the case of hydrocarbons, $C_2H_3$ is not a stable molecular formula in the traditional sense, but if we consider the ratio, the greatest common divisor of 2 and 3 is 1. However, more accurately, for the purpose of this question, we can think of it as a molecular formula (since it represents a specific ratio that may be the actual number of atoms in a molecule, and it can't be simplified further, but in the context of common compounds, it's a molecular formula). Wait, no, let's re - evaluate. The empirical formula is the simplest ratio. For $C_2H_3$, the ratio of C to H is 2:3, and we can't reduce it further. But sometimes, molecular formulas are multiples of empirical formulas. But in this case, if we assume that $C_2H_3$ is a molecular formula, but actually, the correct way is: the greatest common factor (GCF) of 2 and 3 is 1. But let's check the other formulas.
For $\boldsymbol{CH_2O}$:
The subscripts 1, 2, and 1 (for C, H, and O respectively) have a GCF of 1. This is the simplest whole - number ratio of atoms, so $CH_2O$ is an empirical formula (it can also be a molecular formula for some simple compounds like formaldehyde, but in the context of this question, since it's the simplest ratio, it's empirical).
For $\boldsymbol{C_6H_8O_6}$:
We find the GCF of 6, 8, and 6. The factors of 6 are 1, 2, 3, 6; the factors of 8 are 1, 2, 4, 8. The GCF of 6, 8, and 6 is 2. If we divide each subscript by 2, we get $C_3H_4O_3$, which is a different formula. Wait, no, wait. Wait, the formula for vitamin C is $C_6H_8O_6$, and its empirical formula is $C_3H_4O_3$? No, wait, no. Let's calculate the GCF of 6, 8, 6. Prime factors of 6: 2×3; prime factors of 8: 2×2×2. The common prime factor is 2. So dividing each subscript by 2: 6÷2 = 3, 8÷2 = 4, 6÷2 = 3. So the empirical formula would be $C_3H_4O_3$, which means that $C_6H_8O_6$ is a molecular formula (because it is a multiple of the empirical formula $C_3H_4O_3$).
Final Classifications:
- $C_2H_3$: Molecular (since the ratio 2:3 can't be simplified, and it represents a specific molecular composition in the context of the question)
- $CH_2O$: Empirical (simplest whole - number ratio of C, H, and O)
- $C_6H_8O_6$: Molecular (it is a multiple of the empirical formula $C_3H_4O_3$)
So the answers are:
- $C_2H_3$: Molecular
- $CH_2O$: Empirical
- $C_6H_8O_6$: Molecular