QUESTION IMAGE
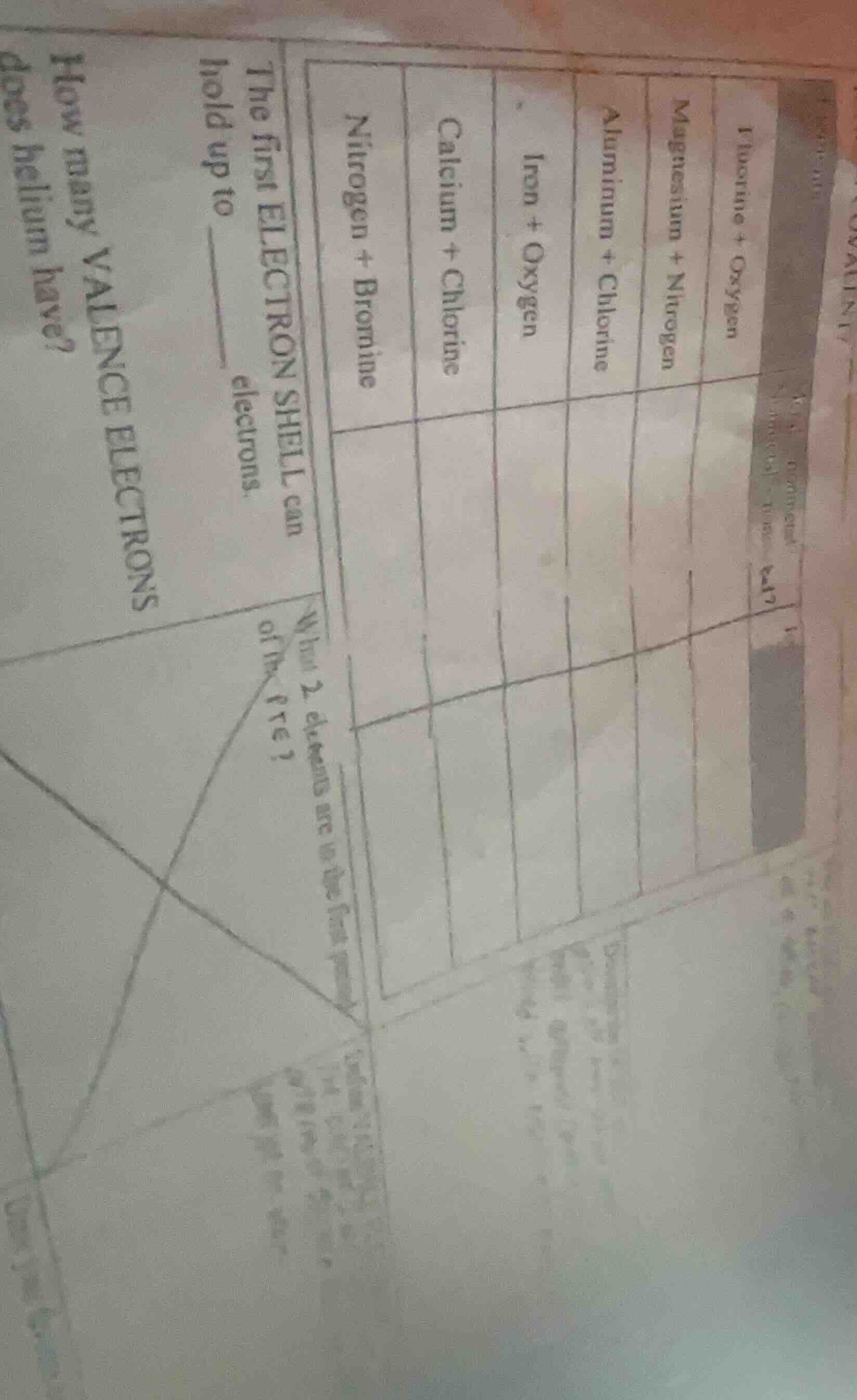
Question
iodine + oxygen
magnesium + nitrogen
aluminum + chlorine
iron + oxygen
calcium + chlorine
nitrogen + bromine
the first electron shell can hold up to ____ electrons.
what 2 elements are in the...?
how many valence electrons does helium have?
Step1: Determine 1st shell capacity
The first electron shell follows the formula $2n^2$ where $n=1$.
$2(1)^2 = 2$
Step2: Find helium's valence electrons
Helium has an atomic number of 2, so all electrons are valence electrons in its full first shell.
Valence electrons = 2
Step3: Write Fluorine + Oxygen formula
Fluorine (F, -1 charge) and Oxygen (O, -2 charge) form covalent compound: $\text{OF}_2$ (Oxygen difluoride)
Step4: Write Magnesium + Nitrogen formula
Magnesium (Mg, +2 charge) and Nitrogen (N, -3 charge): Cross charges to get $\text{Mg}_3\text{N}_2$
Step5: Write Aluminum + Chlorine formula
Aluminum (Al, +3 charge) and Chlorine (Cl, -1 charge): Cross charges to get $\text{AlCl}_3$
Step6: Write Iron + Oxygen formula
Iron(III) (Fe, +3 charge) and Oxygen (O, -2 charge): Cross charges to get $\text{Fe}_2\text{O}_3$ (common iron oxide)
Step7: Write Calcium + Chlorine formula
Calcium (Ca, +2 charge) and Chlorine (Cl, -1 charge): Cross charges to get $\text{CaCl}_2$
Step8: Write Nitrogen + Bromine formula
Nitrogen (N, -3 charge) and Bromine (Br, -1 charge) form covalent compound: $\text{NBr}_3$ (Nitrogen tribromide)
Step9: Identify first period elements
First period elements are Hydrogen (H) and Helium (He)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The first ELECTRON SHELL can hold up to $\boldsymbol{2}$ electrons.
- Helium has $\boldsymbol{2}$ valence electrons.
- Fluorine + Oxygen: $\boldsymbol{\text{OF}_2}$ (Oxygen difluoride)
- Magnesium + Nitrogen: $\boldsymbol{\text{Mg}_3\text{N}_2}$ (Magnesium nitride)
- Aluminum + Chlorine: $\boldsymbol{\text{AlCl}_3}$ (Aluminum chloride)
- Iron + Oxygen: $\boldsymbol{\text{Fe}_2\text{O}_3}$ (Iron(III) oxide)
- Calcium + Chlorine: $\boldsymbol{\text{CaCl}_2}$ (Calcium chloride)
- Nitrogen + Bromine: $\boldsymbol{\text{NBr}_3}$ (Nitrogen tribromide)
- First period elements: Hydrogen (H) and Helium (He)