QUESTION IMAGE
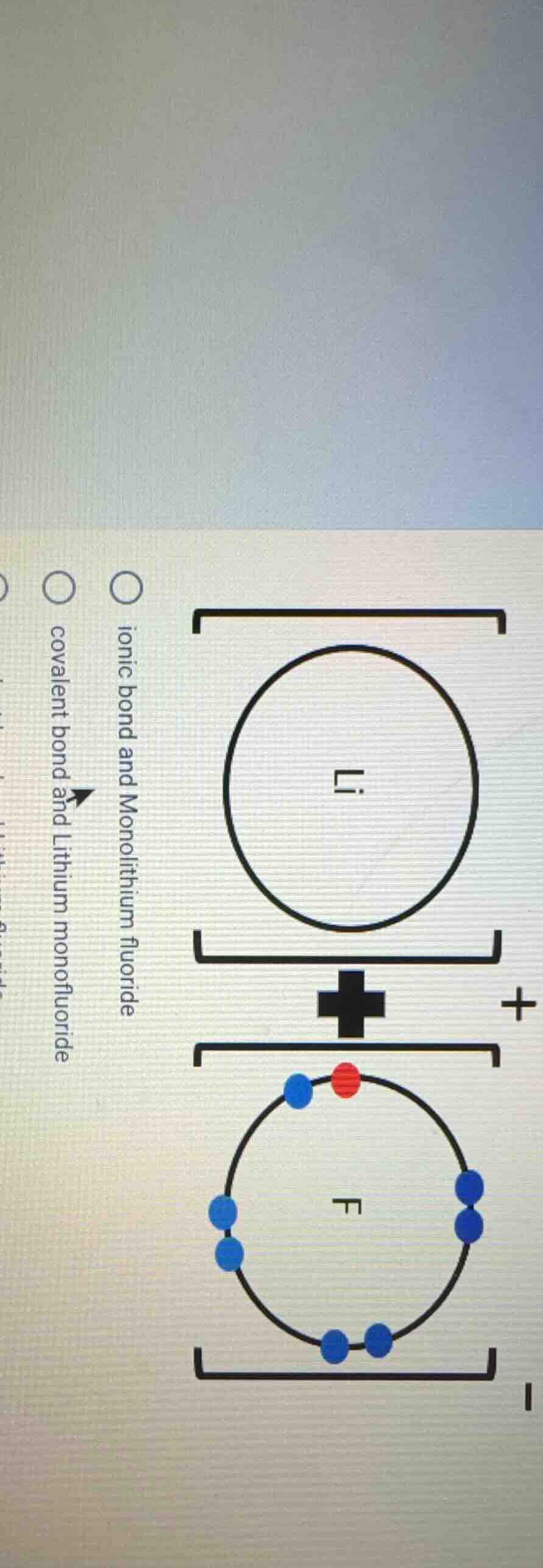
Question
○ ionic bond and monolithium fluoride
○ covalent bond and lithium monofluoride
Lithium (Li) is a group 1 metal that loses its single valence electron to form a positively charged ion ($Li^+$), while fluorine (F) is a group 17 nonmetal that gains this electron to form a negatively charged ion ($F^-$). The electrostatic attraction between these oppositely charged ions forms an ionic bond, and the compound formed is lithium monofluoride (LiF). A covalent bond involves shared electrons, which does not occur here.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
O ionic bond and Monolithium fluoride