QUESTION IMAGE
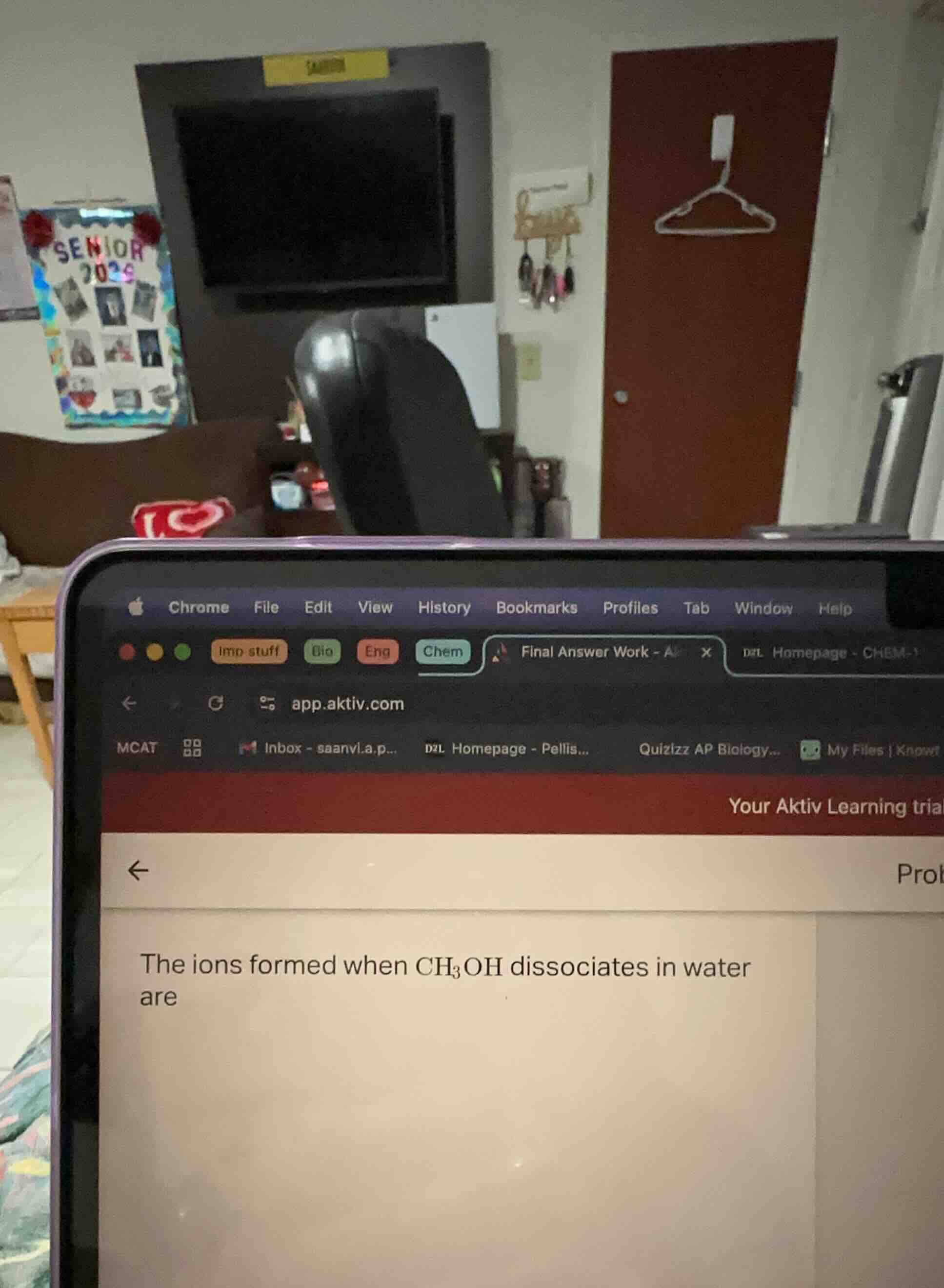
Question
the ions formed when $\ce{ch_{3}oh}$ dissociates in water are
Step1: Identify compound properties
$\text{CH}_3\text{OH}$ (methanol) is a polar covalent compound with a hydroxyl group, but it is a very weak acid. It does not dissociate significantly in water under normal conditions. However, in the context of acid-base chemistry, the extremely limited dissociation produces:
Step2: Write dissociation reaction
The $\text{H}^+$ ion will immediately associate with water to form the hydronium ion:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Methanol dissociates only very slightly in water, forming methoxide ions ($\text{CH}_3\text{O}^-$) and hydronium ions ($\text{H}_3\text{O}^+$) (or equivalently, hydrogen ions $\text{H}^+$ that quickly hydrate to $\text{H}_3\text{O}^+$). For most practical purposes, methanol is considered to remain molecular in water, but its minimal dissociation produces these ions.