QUESTION IMAGE
Question
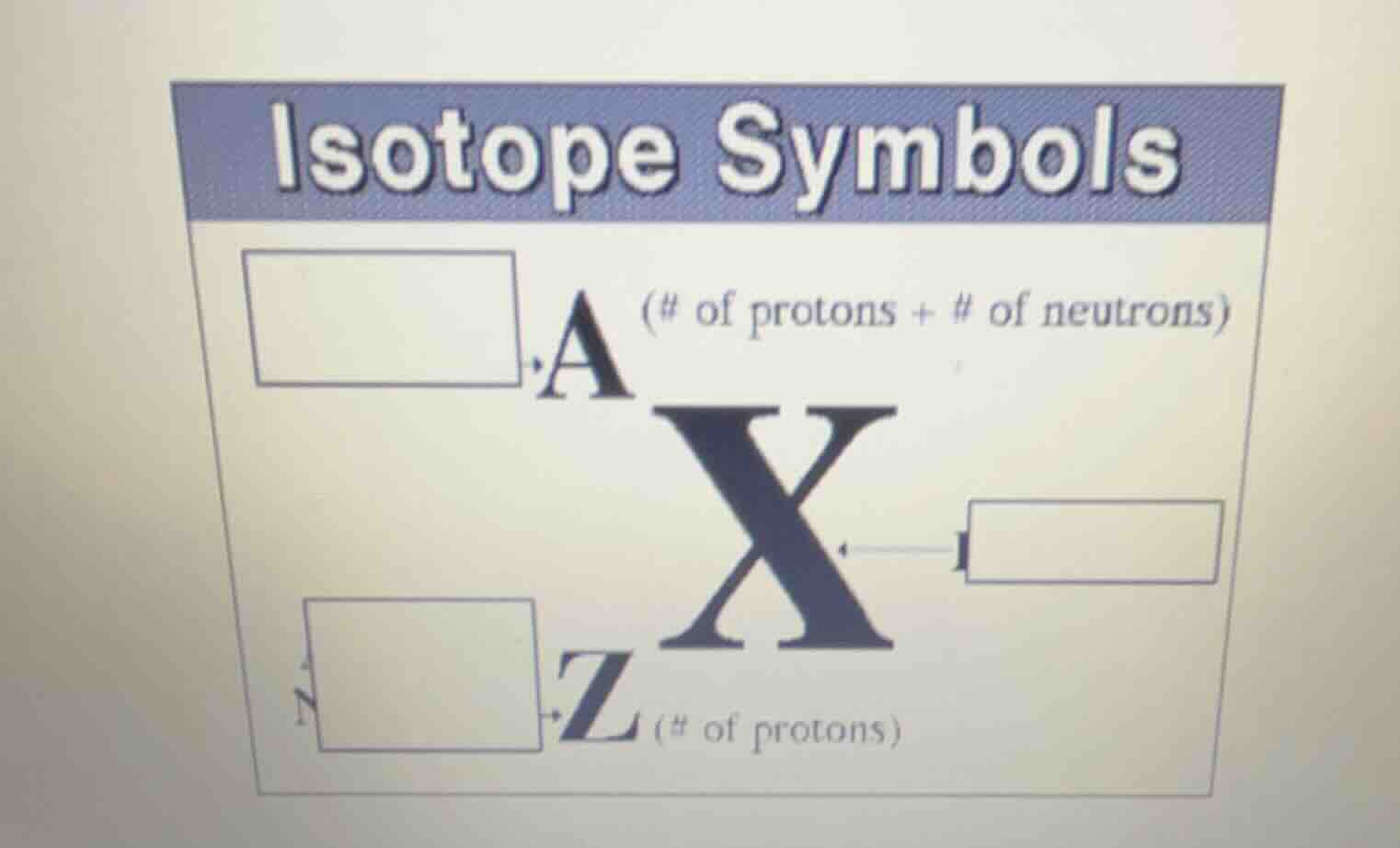
isotope symbols
a (# of protons + # of neutrons)
x
z (# of protons)
Brief Explanations
- The box next to A (mass number) is labeled with the definition of mass number: total number of protons and neutrons in the nucleus.
- The box next to X is the chemical symbol of the element, which identifies the specific element.
- The box next to Z (atomic number) is labeled with the definition of atomic number: number of protons in the nucleus, and since $N = A - Z$, it can also be noted that this box corresponds to the count of protons, with neutrons calculated as $N = A - Z$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Box next to A: Mass Number
- Box next to X: Chemical Symbol of the Element
- Box next to Z: Atomic Number (or Number of Protons)