QUESTION IMAGE
Question
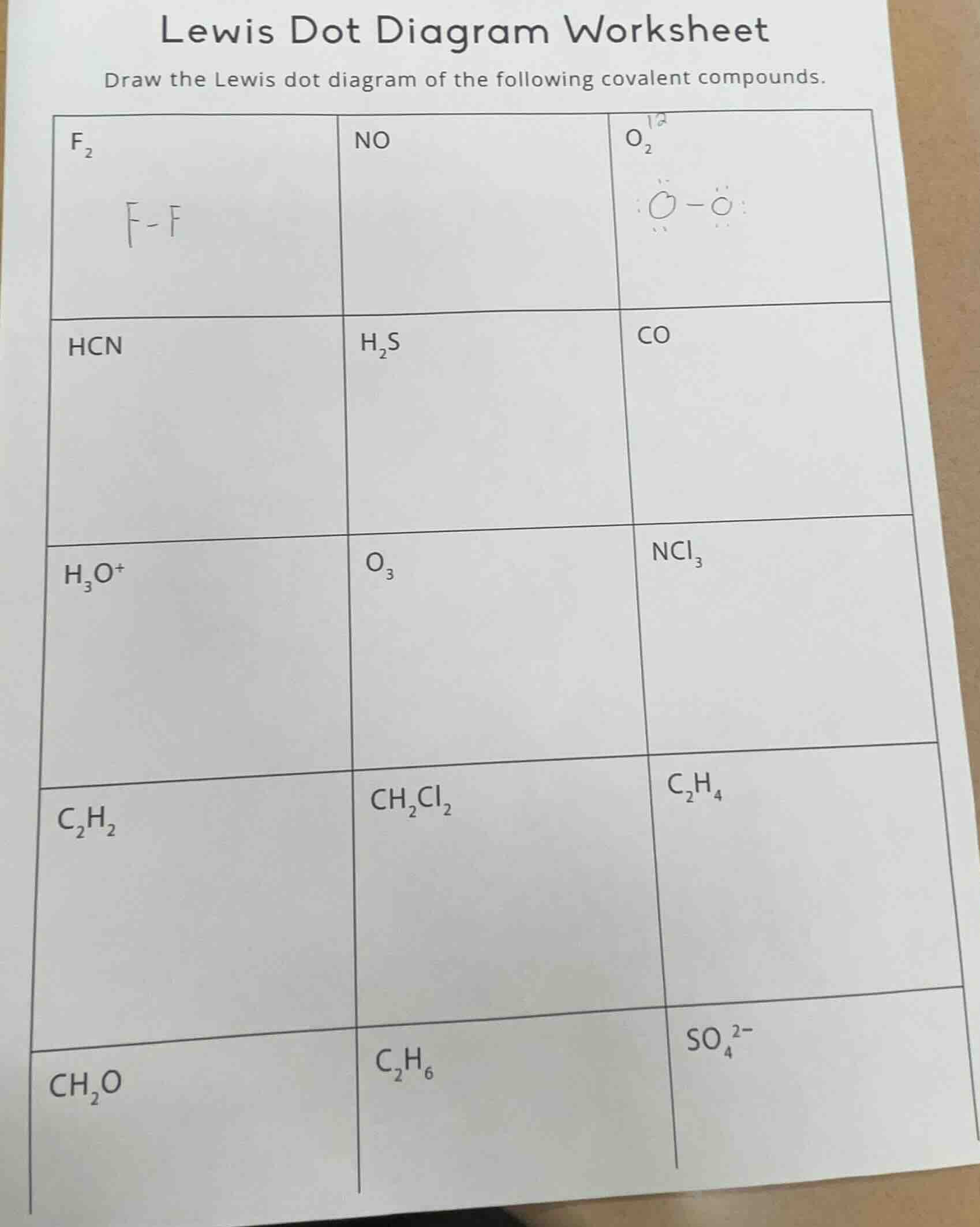
lewis dot diagram worksheet
draw the lewis dot diagram of the following covalent compounds.
f₂\tno\to₂
hcn\th₂s\tco
h₃o⁺\to₃\tncl₃
c₂h₂\tch₂cl₂\tc₂h₄
ch₂o\tc₂h₆\tso₄²⁻
Step1: Calculate total valence electrons for NO
N (5) + O (6) = 11 valence electrons
Step2: Draw NO Lewis structure
$$:\dot{N}=\ddot{O}:$$
(Note: One unpaired electron on N, satisfies octet for O, 7 electrons on N)
Step3: Calculate total valence electrons for HCN
H (1) + C (4) + N (5) = 10 valence electrons
Step4: Draw HCN Lewis structure
$$H-C\equiv N:$$
Step5: Calculate total valence electrons for H₂S
2×H (1) + S (6) = 8 valence electrons
Step6: Draw H₂S Lewis structure
Step7: Calculate total valence electrons for CO
C (4) + O (6) = 10 valence electrons
Step8: Draw CO Lewis structure
$$:C\equiv O:$$
Step9: Calculate total valence electrons for H₃O⁺
3×H (1) + O (6) - 1 (charge) = 8 valence electrons
Step10: Draw H₃O⁺ Lewis structure
Step11: Calculate total valence electrons for O₃
3×O (6) = 18 valence electrons
Step12: Draw O₃ Lewis structure (resonance form)
$$:\ddot{O}-\ddot{O}=\ddot{O}: \longleftrightarrow :\ddot{O}=\ddot{O}-\ddot{O}:$$
Step13: Calculate total valence electrons for NCl₃
N (5) + 3×Cl (7) = 26 valence electrons
Step14: Draw NCl₃ Lewis structure
Step15: Calculate total valence electrons for C₂H₂
2×C (4) + 2×H (1) = 10 valence electrons
Step16: Draw C₂H₂ Lewis structure
$$H-C\equiv C-H$$
Step17: Calculate total valence electrons for CH₂Cl₂
C (4) + 2×H (1) + 2×Cl (7) = 20 valence electrons
Step18: Draw CH₂Cl₂ Lewis structure
Step19: Calculate total valence electrons for C₂H₄
2×C (4) + 4×H (1) = 12 valence electrons
Step20: Draw C₂H₄ Lewis structure
(Each C has two lone pairs implied in octet, shown as:
)
Step21: Calculate total valence electrons for CH₂O
C (4) + 2×H (1) + O (6) = 12 valence electrons
Step22: Draw CH₂O Lewis structure
Step23: Calculate total valence electrons for C₂H₆
2×C (4) + 6×H (1) = 14 valence electrons
Step24: Draw C₂H₆ Lewis structure
Step25: Calculate total valence electrons for SO₄²⁻
S (6) + 4×O (6) + 2 (charge) = 32 valence electrons
Step26: Draw SO₄²⁻ Lewis structure (resonance form)
(One double bond, three single bonds; resonance swaps double bond position)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $F_2$ (completed): $:\ddot{F}-\ddot{F}:$
- $NO$: $:\dot{N}=\ddot{O}:$
- $O_2$ (completed, corrected): $:\ddot{O}=\ddot{O}:$
- $HCN$: $H-C\equiv N:$
- $H_2S$:
- $CO$: $:C\equiv O:$
- $H_3O^+$:
- $O_3$: $:\ddot{O}-\ddot{O}=\ddot{O}: \longleftrightarrow :\ddot{O}=\ddot{O}-\ddot{O}:$
- $NCl_3$:
- $C_2H_2$: $H-C\equiv C-H$
- $CH_2Cl_2$:
- $C_2H_4$:
- $CH_2O$:
- $C_2H_6$:
- $SO_4^{2-}$:
(resonance structures exist)