QUESTION IMAGE
Question
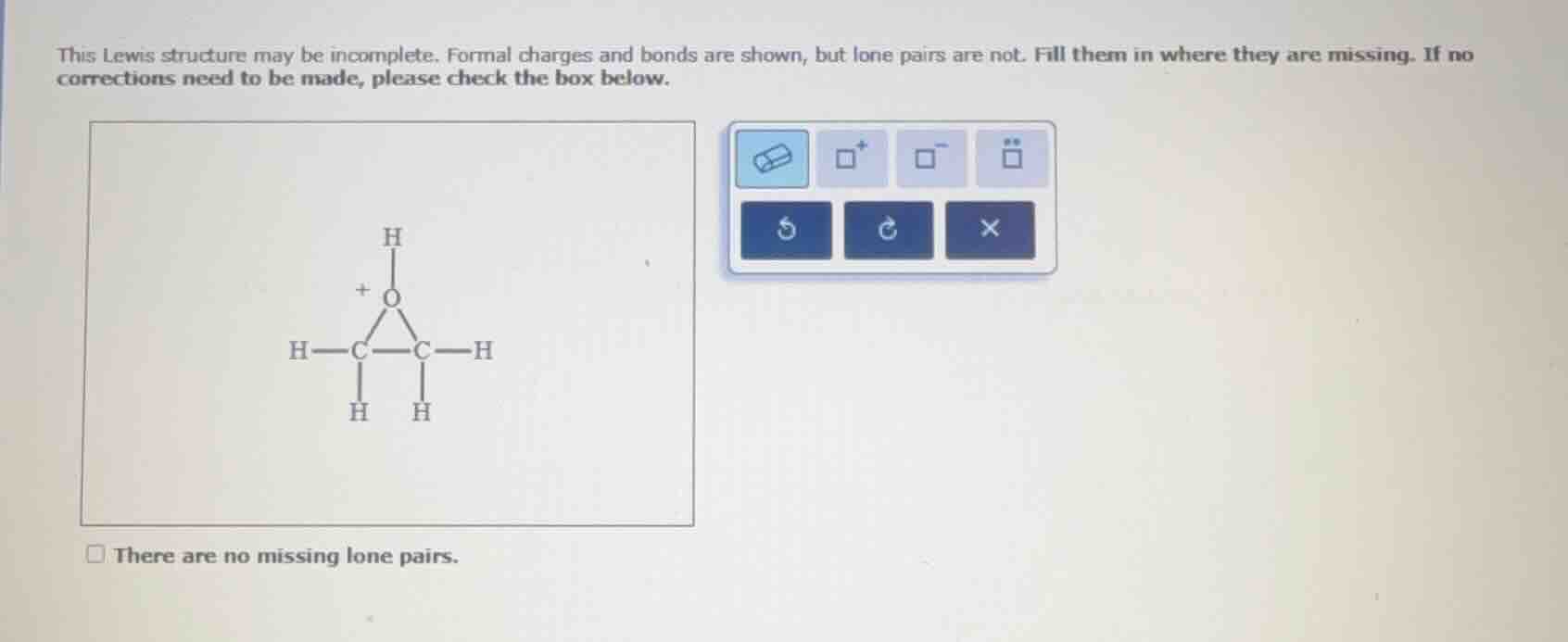
this lewis structure may be incomplete. formal charges and bonds are shown, but lone pairs are not. fill them in where they are missing. if no corrections need to be made, please check the box below.
there are no missing lone pairs.
Step1: Analyze the Oxygen Atom
Oxygen has 6 valence electrons. In the structure, it has a formal charge of +1 and is bonded to two carbon atoms and one hydrogen atom (3 bonds, each contributing 1 electron to its valence shell from bonding). So, the number of lone pairs: Valence electrons - (bonds + formal charge) = 6 - (3 + 1) = 2? Wait, no, formal charge formula is \( \text{Formal Charge} = \text{Valence Electrons} - (\text{Non - bonding Electrons} + \frac{1}{2}\text{Bonding Electrons}) \). For O with +1 charge: \( + 1=6-(N + \frac{1}{2}\times3) \), solving for N (non - bonding electrons, which are lone pair electrons, each lone pair is 2 electrons). So \( N=6 - \frac{3}{2}-1=\frac{12 - 3 - 2}{2}=\frac{7}{2} \)? Wait, maybe a better approach: Carbon atoms: each carbon is bonded to 4 groups (so they have 4 bonds, no lone pairs, since carbon has 4 valence electrons and all are in bonding). Hydrogen has 1 bond, no lone pairs. Oxygen: Let's count the bonds. The O is in a three - membered ring with two C atoms and bonded to H. So O has 3 bonds (2 with C, 1 with H). Valence electrons for O: 6. Formal charge is +1, so \( \text{Formal Charge}=6 - (\text{lone pair electrons}+\frac{\text{bonding electrons}}{2}) \). Bonding electrons: 3 bonds × 2 electrons per bond = 6 (but each bond is shared, so for O, bonding electrons are 3 (since each bond contributes 1 electron to O's count? Wait, no, in a covalent bond, each atom contributes one electron. So for a single bond, O has 1 electron from each bond. So 3 bonds mean 3 bonding electrons (from O's side). Then \( + 1=6-(L + 3) \), where L is the number of lone pair electrons (each lone pair is 2 electrons, so the number of lone pairs is \( \frac{L}{2} \)). So \( L=6 - 3 - 1 = 2 \). So lone pair electrons are 2, which is 1 lone pair? Wait, no, 2 electrons is 1 lone pair? Wait, no, a lone pair is 2 electrons. So if L = 2, that's 1 lone pair? Wait, I think I made a mistake. Let's re - calculate. The correct formula: \( \text{Formal Charge} = V - (N + B) \), where V is valence electrons (6 for O), N is number of non - bonding electrons (lone pair electrons, each lone pair is 2 electrons, so N = 2×number of lone pairs), and B is the number of bonding electrons (each bond has 2 electrons, so for O, in 3 bonds, B = 3 (since each bond is a single bond, O contributes 1 electron to each bond, so total bonding electrons from O's side is 3). Wait, I'm getting confused. Let's look at the structure again. The two carbon atoms: each C is bonded to 4 atoms (so they have 4 single bonds, no lone pairs, as carbon follows octet (4 bonds, 8 electrons, 4 from bonding, 4 from? Wait, no, carbon has 4 valence electrons, and in 4 single bonds, it has 8 electrons (4 bonds × 2 electrons), which satisfies the octet. So C has no lone pairs. Hydrogen has 1 bond, no lone pairs. Oxygen: Let's use the octet rule (except for formal charge). O with +1 charge: it has less electrons. O normally has 6 valence electrons, with +1 charge, it has 5 electrons in its valence shell (since formal charge is loss of 1 electron). The bonding electrons: 3 bonds (so 3 electrons from bonding, since each bond is a single bond, O shares 1 electron in each bond). So total electrons in O's valence shell: 3 (bonding) + lone pair electrons. This should equal 5 (because of +1 charge, 6 - 1 = 5). So lone pair electrons = 5 - 3 = 2. So that's 1 lone pair (2 electrons). Wait, but the button for lone pairs is a pair of two dots. So we need to add 1 lone pair (two dots) to the oxygen atom.
Step2: Add Lone Pairs to Oxygen
So the oxygen atom in the structu…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Add one lone pair (two dots) to the oxygen atom. The carbon and hydrogen atoms have no missing lone pairs. So the Lewis structure should have a lone pair on the oxygen.