QUESTION IMAGE
Question
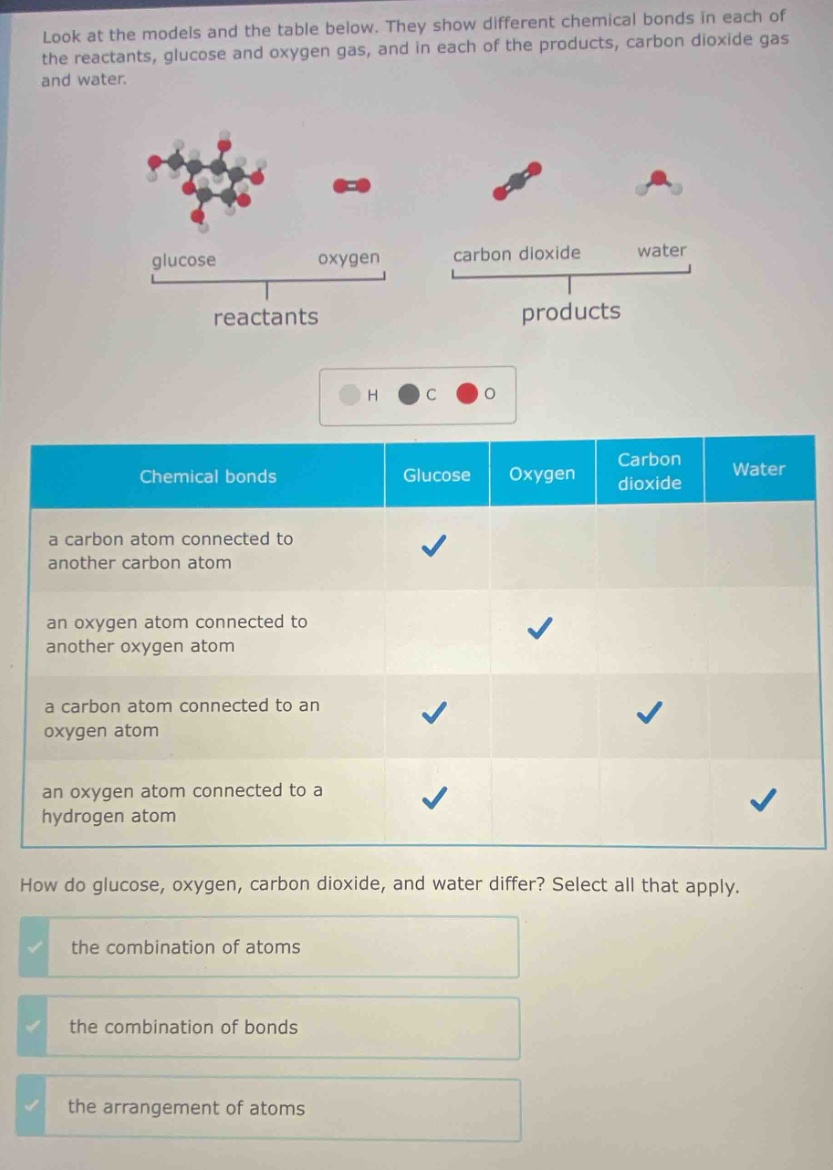
look at the models and the table below. they show different chemical bonds in each of the reactants, glucose and oxygen gas, and in each of the products, carbon dioxide gas and water.
glucose, oxygen, carbon dioxide, water models with legend: h (gray), c (black), o (red)
table with chemical bonds (a carbon atom connected to another carbon atom; an oxygen atom connected to another oxygen atom; a carbon atom connected to an oxygen atom; an oxygen atom connected to a hydrogen atom) and columns glucose, oxygen, carbon dioxide, water with checkmarks
how do glucose, oxygen, carbon dioxide, and water differ? select all that apply.
- the combination of atoms (checked)
- the combination of bonds (checked)
- the arrangement of atoms (checked)
- Combination of atoms: Glucose has C, H, O; Oxygen has only O; Carbon dioxide has C and O; Water has H and O. So their atom combinations differ.
- Combination of bonds: From the table, glucose has C - C, C - O, O - H bonds; oxygen has O - O; carbon dioxide has C - O; water has O - H. Their bond combinations differ.
- Arrangement of atoms: Different molecules (glucose, O₂, CO₂, H₂O) have different atomic arrangements (e.g., glucose is a complex organic molecule, O₂ is diatomic, CO₂ is linear, H₂O is bent).
All three options are correct as the substances differ in atom combination (types of atoms), bond combination (types of bonds present), and atom arrangement (structure of molecules).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- the combination of atoms
- the combination of bonds
- the arrangement of atoms