QUESTION IMAGE
Question
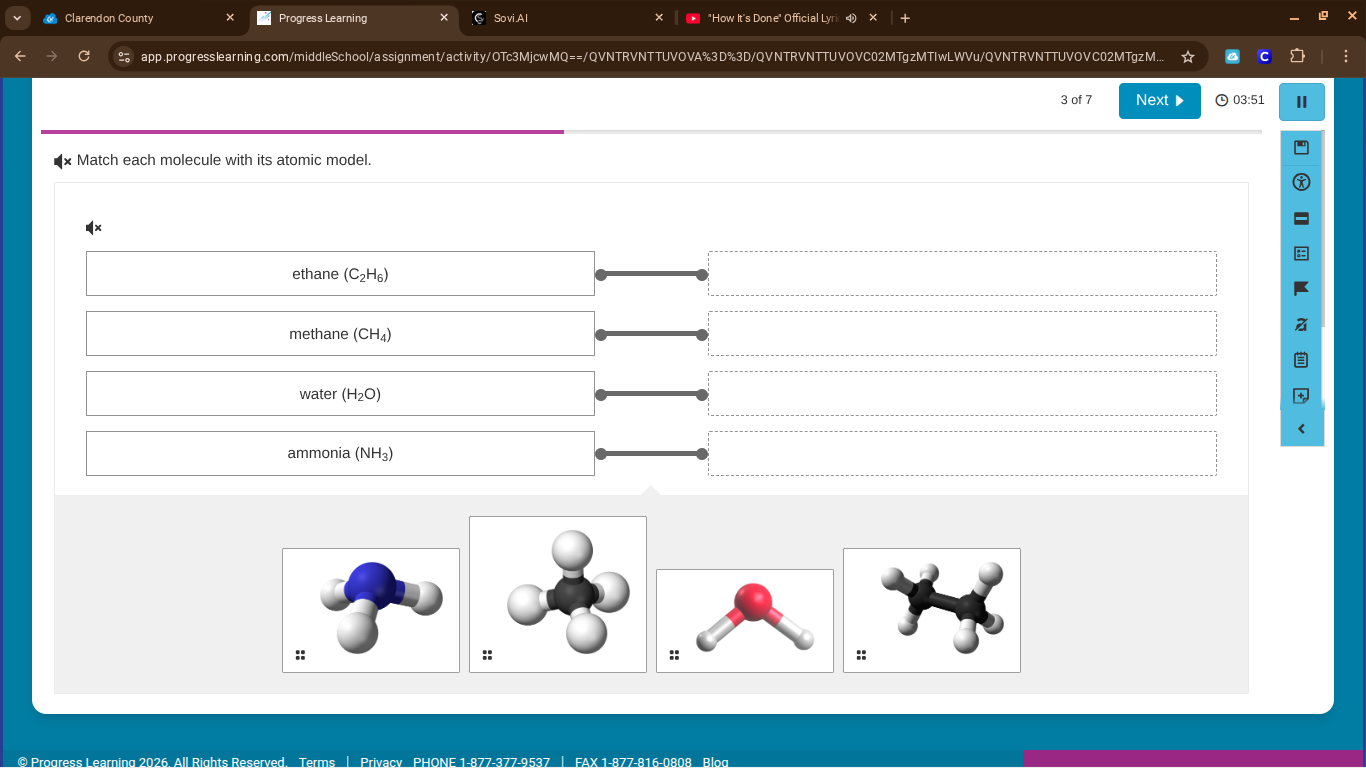
match each molecule with its atomic model.
ethane (c₂h₆)
methane (ch₄)
water (h₂o)
ammonia (nh₃)
Brief Explanations
To solve this, we analyze the molecular structures:
- Ethane (\(C_2H_6\)): Has two carbon atoms bonded in a chain with six hydrogens. The last model (two black carbons, six white hydrogens) matches.
- Methane (\(CH_4\)): Tetrahedral structure (one black carbon, four white hydrogens), the second model.
- Water (\(H_2O\)): Bent structure (one red oxygen, two white hydrogens), the third model.
- Ammonia (\(NH_3\)): Trigonal pyramidal (one blue nitrogen, three white hydrogens), the first model.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- ethane (\(C_2H_6\)) → last model (two black carbons, six white hydrogens)
- methane (\(CH_4\)) → second model (one black carbon, four white hydrogens)
- water (\(H_2O\)) → third model (one red oxygen, two white hydrogens)
- ammonia (\(NH_3\)) → first model (one blue nitrogen, three white hydrogens)