QUESTION IMAGE
Question
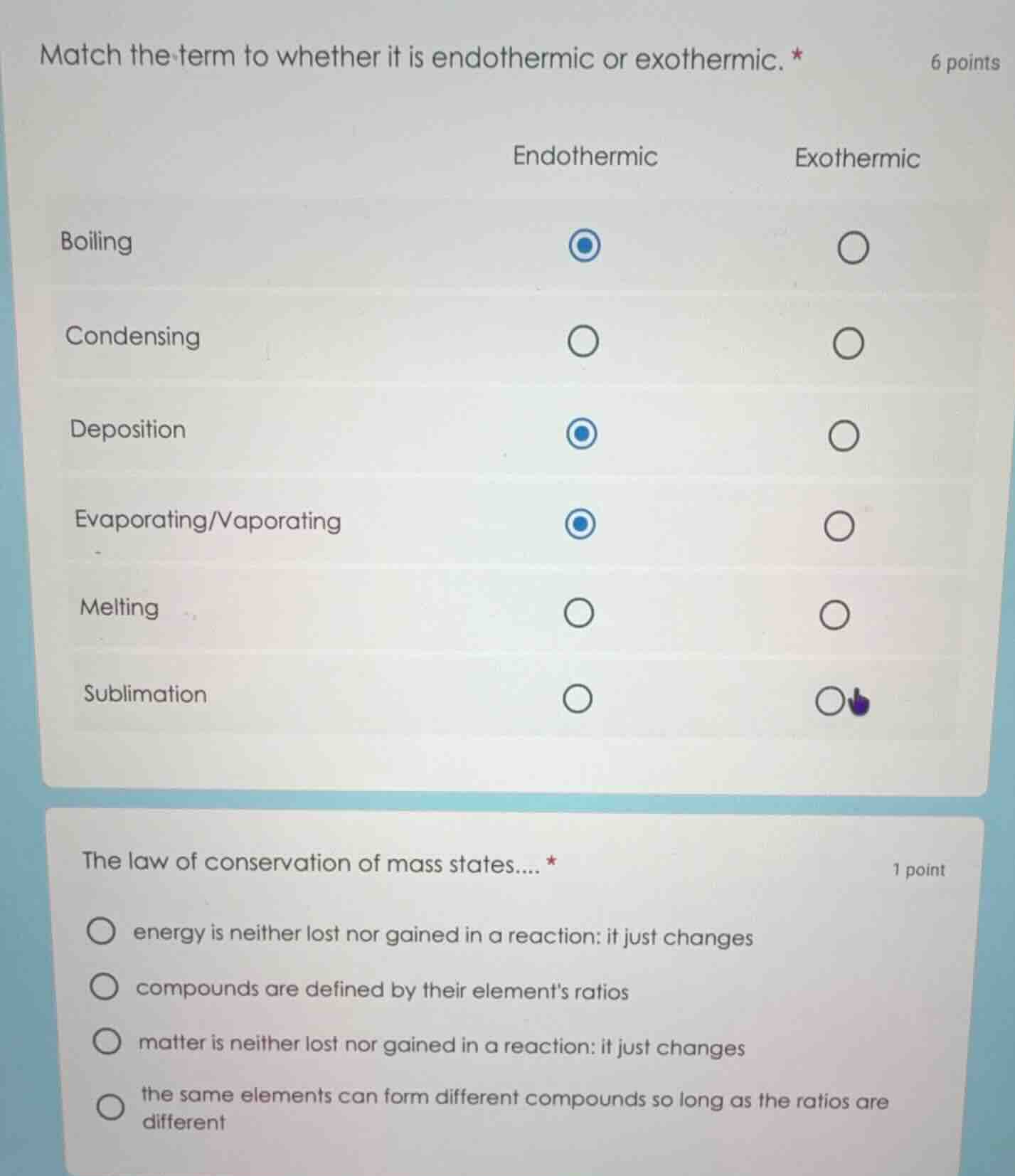
match the term to whether it is endothermic or exothermic. * 6 points
endothermic exothermic
boiling
condensing
deposition
evaporating/vaporating
melting
sublimation
the law of conservation of mass states.... * 1 point
energy is neither lost nor gained in a reaction: it just changes
compounds are defined by their elements ratios
matter is neither lost nor gained in a reaction: it just changes
the same elements can form different compounds so long as the ratios are different
First Part (Phase Changes - Endothermic/Exothermic)
- Boiling: Correctly marked as Endothermic (needs heat to convert liquid to gas).
- Condensing: Exothermic (gas to liquid, releases heat).
- Deposition: Exothermic (gas to solid, releases heat; was incorrectly marked as Endothermic).
- Evaporating/Vaporating: Correctly marked as Endothermic (liquid to gas, needs heat).
- Melting: Endothermic (solid to liquid, needs heat; was unmarked).
- Sublimation: Endothermic (solid to gas, needs heat; was incorrectly marked as Exothermic).
Second Part (Law of Conservation of Mass)
The law of conservation of mass states that in a chemical reaction, matter (mass) is neither created nor destroyed—only rearranged.
- Option 1: Describes conservation of energy (not mass).
- Option 2: Describes definite proportions (not conservation of mass).
- Option 3: Correctly states matter is neither lost nor gained, just changed.
- Option 4: Describes multiple proportions (not conservation of mass).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
Phase Changes (Correct Selections):
- Boiling: Endothermic (correct as marked).
- Condensing: Exothermic.
- Deposition: Exothermic.
- Evaporating/Vaporating: Endothermic (correct as marked).
- Melting: Endothermic.
- Sublimation: Endothermic.
Law of Conservation of Mass:
The correct option is:
C. matter is neither lost nor gained in a reaction: it just changes