QUESTION IMAGE
Question
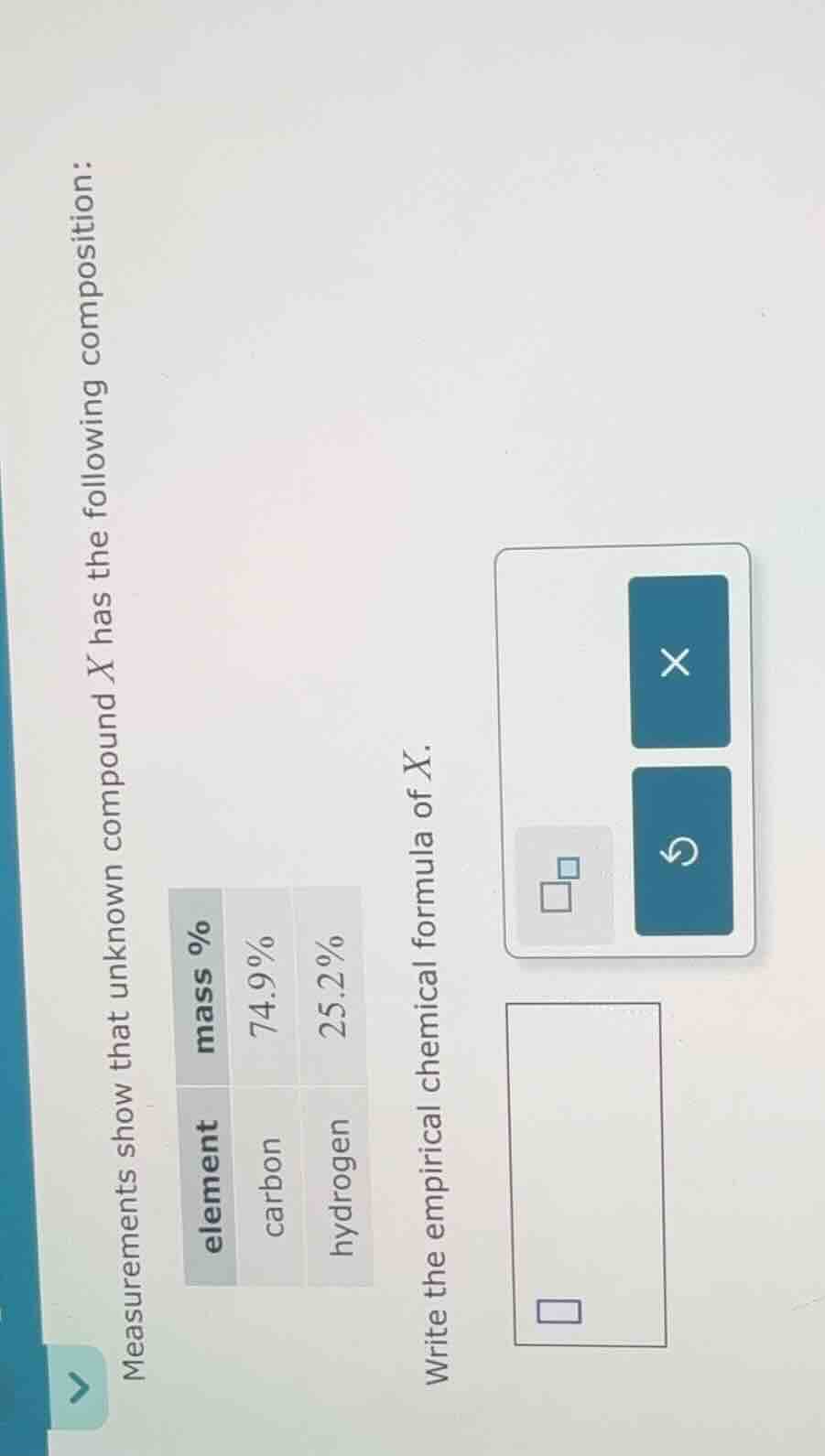
measurements show that unknown compound x has the following composition: element | mass % carbon | 74.9% hydrogen | 25.2% write the empirical chemical formula of x.
Step1: Assume 100g of compound X
Mass of C = 74.9g, Mass of H = 25.2g.
Step2: Calculate moles of each element
Moles of C: $\frac{74.9\mathrm{g}}{12.01\mathrm{g/mol}} \approx 6.24\mathrm{mol}$
Moles of H: $\frac{25.2\mathrm{g}}{1.008\mathrm{g/mol}} \approx 25.0\mathrm{mol}$
Step3: Find mole ratio
Divide by smallest moles (6.24):
Ratio of C: $\frac{6.24}{6.24} = 1$
Ratio of H: $\frac{25.0}{6.24} \approx 4$
So, empirical formula is $CH_4$? Wait, wait, recalculate H moles: 25.2 / 1.008 = 25? Wait 1.00825 = 25.2, yes. And C: 74.9/12.01 ≈ 6.24? Wait no, 12.016 = 72.06, 74.9 -72.06=2.84, 2.84/12.01≈0.236, so total C moles≈6.236? Wait, maybe I miscalculated. Wait 74.9 /12.01: 12*6=72, 74.9-72=2.9, 2.9/12≈0.24, so 6.24. H:25.2/1.008=25. So ratio C:H = 6.24:25. Divide by 6.24: C=1, H≈25/6.24≈4.01, so ~4. So empirical formula is $CH_4$? Wait no, wait 74.9% C and 25.2% H. Let's check mass of CH4: C is 12, H is 4, total 16. C%: 12/16=75%, H%:4/16=25%. Oh! 74.9 is close to 75, 25.2 close to 25. So the empirical formula is $CH_4$. Wait, maybe my initial mole calculation had a rounding error. Let's do it precisely:
Moles of C: 74.9 / 12.011 ≈ 6.236
Moles of H: 25.2 / 1.008 ≈ 25.0
Ratio C:H = 6.236 : 25.0
Divide by 6.236:
C: 1
H: 25.0 / 6.236 ≈ 4.009 ≈ 4
So the empirical formula is $CH_4$. Wait, but let's check the mass percentages. For $CH_4$:
Mass of C: 12.01, Mass of H: 4.032 (since H is 1.008*4=4.032)
Total mass: 12.01 + 4.032 = 16.042
C%: (12.01 /16.042)*100 ≈ 74.87%, which is ~74.9%
H%: (4.032 /16.042)*100 ≈25.13%, ~25.2%. Perfect. So the empirical formula is $CH_4$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boxed{CH_4}$