QUESTION IMAGE
Question
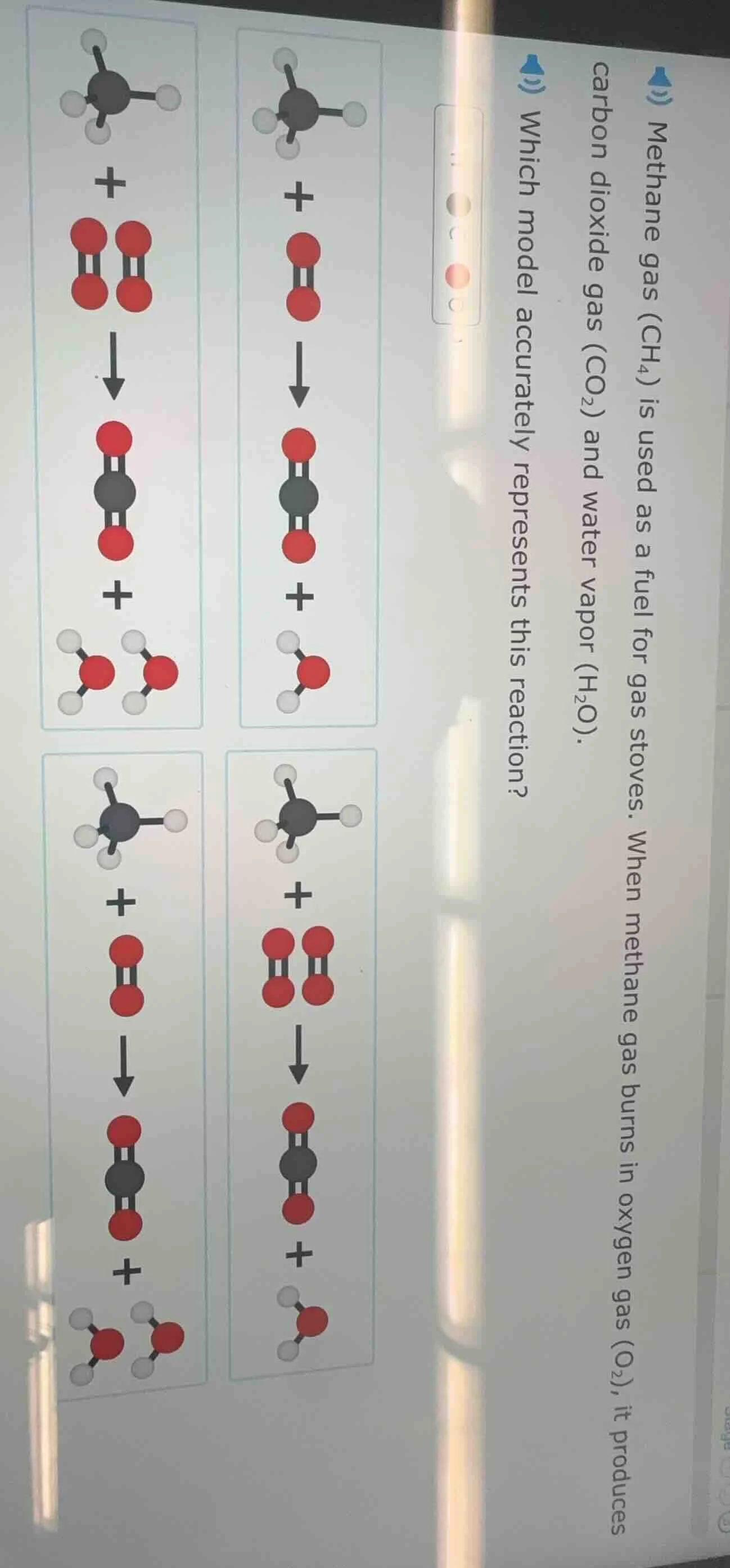
methane gas ($ch_4$) is used as a fuel for gas stoves. when methane gas burns in oxygen gas ($o_2$), it produces carbon dioxide gas ($co_2$) and water vapor ($h_2o$). which model accurately represents this reaction?
Step1: Write balanced reaction
The combustion of methane follows:
Step2: Match to particle model
Count reactants/products: 1 $\text{CH}_4$ (1 black, 4 white), 2 $\text{O}_2$ (4 red, paired as 2 molecules), 1 $\text{CO}_2$ (1 black, 2 red), 2 $\text{H}_2\text{O}$ (2 red, 4 white, paired as 2 molecules). This matches the top-left model.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The top-left model (1 methane molecule + 2 oxygen molecules → 1 carbon dioxide molecule + 2 water molecules)