QUESTION IMAGE
Question
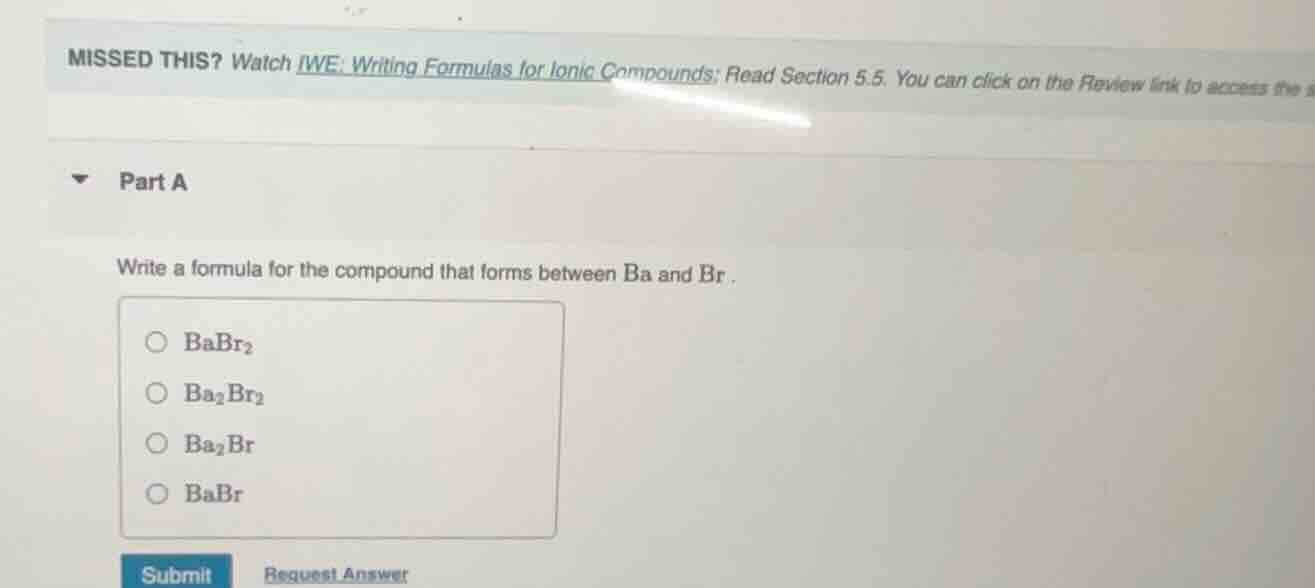
missed this? watch iwe: writing formulas for ionic compounds; read section 5.5. you can click on the review link to access the part a write a formula for the compound that forms between ba and br. babr₂ ba₂br₂ ba₂br babr
To determine the formula of the ionic compound formed between Barium (Ba) and Bromine (Br), we first identify the charges of the ions. Barium is an alkaline earth metal, so it forms a \( Ba^{2+} \) ion (losing 2 electrons). Bromine is a halogen and forms a \( Br^- \) ion (gaining 1 electron). For the compound to be electrically neutral, the total positive charge must equal the total negative charge. Let the number of \( Ba^{2+} \) ions be \( x \) and the number of \( Br^- \) ions be \( y \). Then, \( 2x = y \) (since the charge of \( Ba^{2+} \) is +2 and \( Br^- \) is -1). The simplest ratio that satisfies this is \( x = 1 \) and \( y = 2 \), so the formula is \( BaBr_2 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \boldsymbol{BaBr_2} \)