QUESTION IMAGE
Question
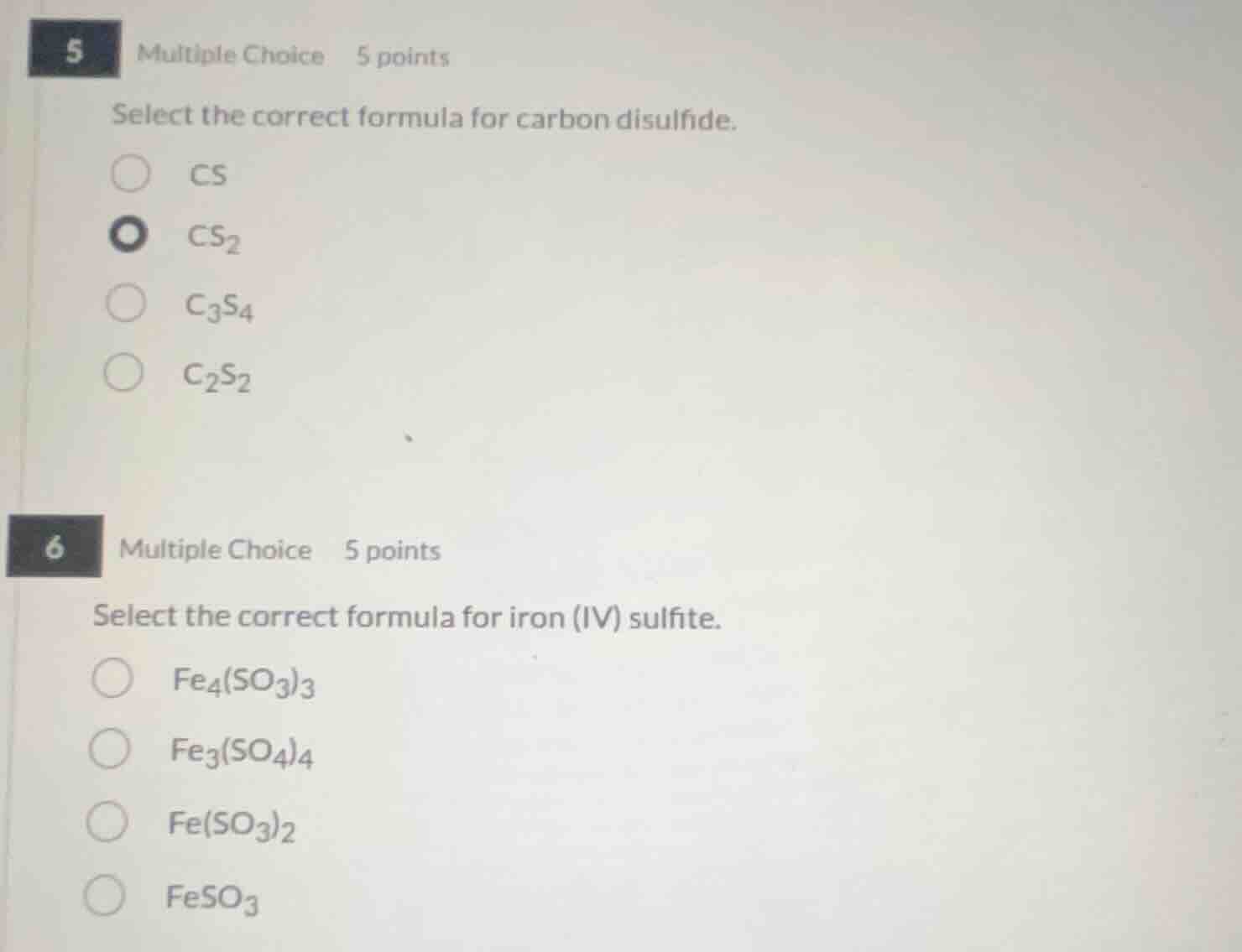
5 multiple choice 5 points
select the correct formula for carbon disulfide.
cs
cs₂
c₃s₄
c₂s₂
6 multiple choice 5 points
select the correct formula for iron (iv) sulfite.
fe₄(so₃)₃
fe₃(so₄)₄
fe(so₃)₂
feso₃
Step1: Analyze carbon disulfide
Carbon is C, "disulfide" means 2 sulfur (S) atoms. Formula: $\text{CS}_2$
Step2: Analyze iron (IV) sulfite
Iron(IV) has charge $+4$, sulfite ion is $\text{SO}_3^{2-}$. Balance charges: $+4 + 2\times(-2)=0$. Formula: $\text{Fe(SO}_3\text{)}_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{CS}_2$
- $\text{Fe(SO}_3\text{)}_2$