QUESTION IMAGE
Question
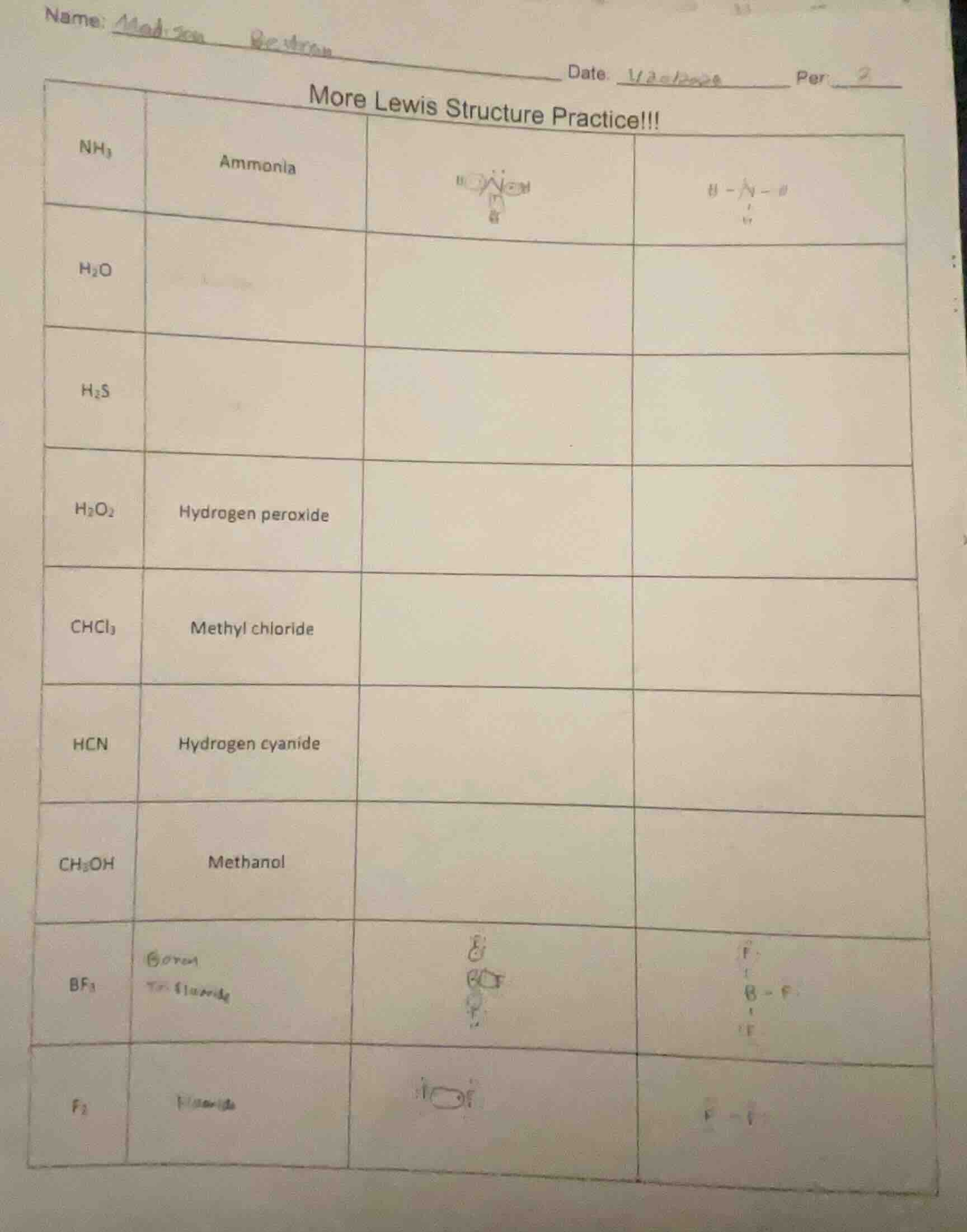
name: madison beutron date: 1/2=12024 per 2 more lewis structure practice!!! nh₃ ammonia h₂o h₂s h₂o₂ hydrogen peroxide chcl₃ methyl chloride hcn hydrogen cyanide ch₃oh methanol bf₃ boron trifluoride f₂ fluoride
To solve for the Lewis structure of \( \ce{H2O} \) (Water), we follow these steps:
Step 1: Determine the total number of valence electrons
- Hydrogen (\( \ce{H} \)) has 1 valence electron, and there are 2 H atoms: \( 2 \times 1 = 2 \)
- Oxygen (\( \ce{O} \)) has 6 valence electrons.
- Total valence electrons: \( 2 + 6 = 8 \)
Step 2: Arrange the atoms
- The central atom is usually the least electronegative atom. Here, oxygen is the central atom, with the two hydrogen atoms bonded to it.
Step 3: Draw single bonds between the central atom and the surrounding atoms
- Each single bond (H - O - H) uses 2 electrons. So, 2 bonds use \( 2 \times 2 = 4 \) electrons.
Step 4: Distribute the remaining electrons as lone pairs on the central atom
- Remaining electrons: \( 8 - 4 = 4 \)
- These 4 electrons form two lone pairs on the oxygen atom.
The Lewis structure of \( \ce{H2O} \) is:
\[
\ce{H - \overset{..}{O} - H}
\]
where the two pairs of dots (..) represent the lone pairs of electrons on the oxygen atom.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the Lewis structure of \( \ce{H2O} \) (Water), we follow these steps:
Step 1: Determine the total number of valence electrons
- Hydrogen (\( \ce{H} \)) has 1 valence electron, and there are 2 H atoms: \( 2 \times 1 = 2 \)
- Oxygen (\( \ce{O} \)) has 6 valence electrons.
- Total valence electrons: \( 2 + 6 = 8 \)
Step 2: Arrange the atoms
- The central atom is usually the least electronegative atom. Here, oxygen is the central atom, with the two hydrogen atoms bonded to it.
Step 3: Draw single bonds between the central atom and the surrounding atoms
- Each single bond (H - O - H) uses 2 electrons. So, 2 bonds use \( 2 \times 2 = 4 \) electrons.
Step 4: Distribute the remaining electrons as lone pairs on the central atom
- Remaining electrons: \( 8 - 4 = 4 \)
- These 4 electrons form two lone pairs on the oxygen atom.
The Lewis structure of \( \ce{H2O} \) is:
\[
\ce{H - \overset{..}{O} - H}
\]
where the two pairs of dots (..) represent the lone pairs of electrons on the oxygen atom.