QUESTION IMAGE
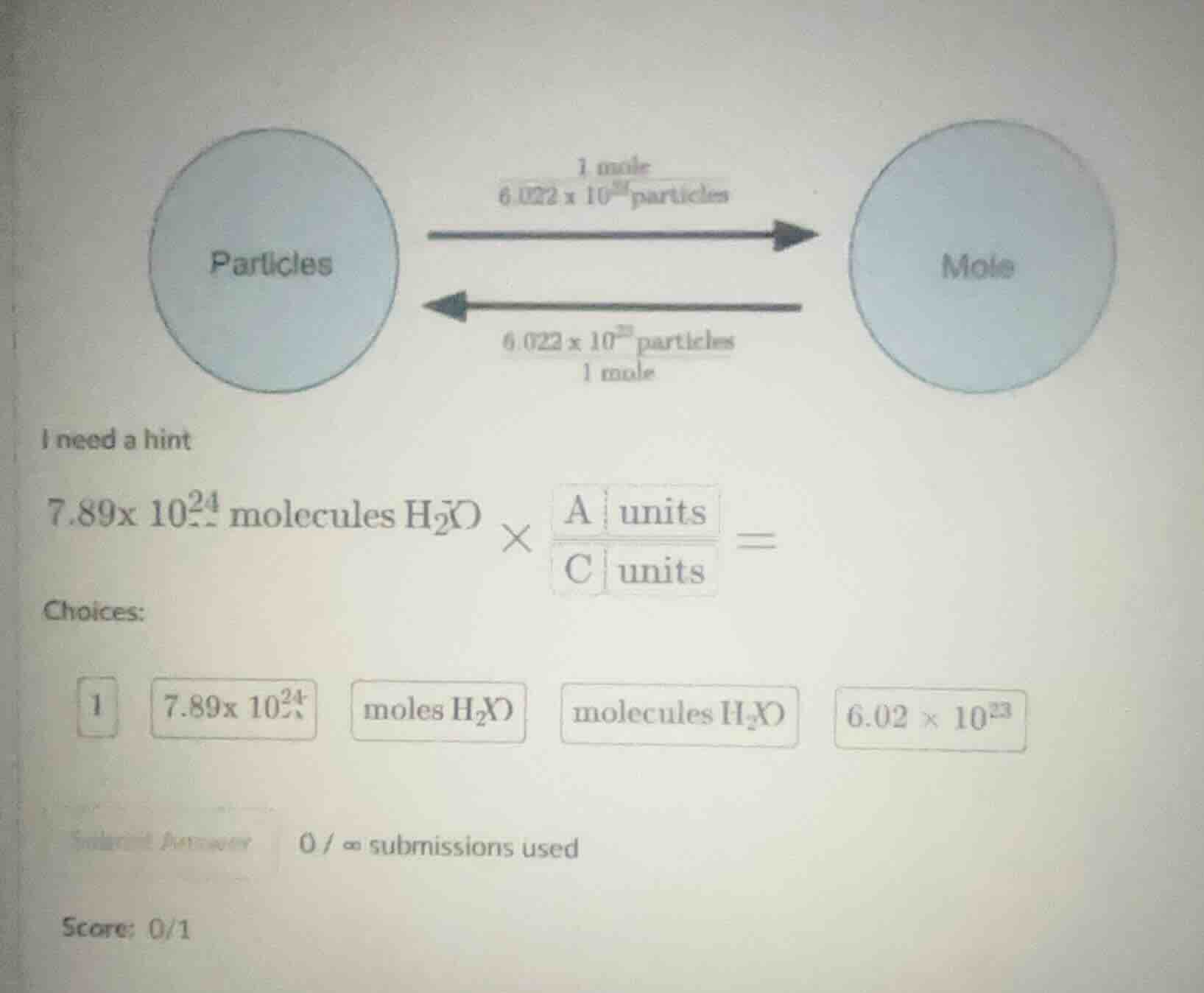
Question
i need a hint
7.89×10²⁴ molecules h₂o × (a units / c units) =
choices:
1 7.89×10²⁴ moles h₂o molecules h₂o 6.02×10²³
submitted answer 0/∞ submissions used
score: 0/1
Step1: Recall the mole - particle conversion factor
The conversion factor between moles and particles (molecules in this case) is based on Avogadro's number, which is \(6.022\times 10^{23}\) particles per mole. To convert from molecules (particles) to moles, we use the conversion factor \(\frac{1\space mole}{6.022\times 10^{23}\space molecules}\) (or \(\frac{1\space mole}{6.02\times 10^{23}\space molecules}\) as an approximation here).
So, for the given problem, we have \(7.89\times 10^{24}\) molecules of \(H_2O\) and we want to convert to moles. The conversion factor should be \(\frac{1\space mole}{6.02\times 10^{23}\space molecules\space H_2O}\) (or \(\frac{1\space mole}{6.022\times 10^{23}\space molecules\space H_2O}\)). Looking at the choices, the numerator (A units) should be \(1\space mole\) (or represented as 1 in terms of the coefficient if we consider the units) and the denominator (C units) should be \(6.02\times 10^{23}\space molecules\space H_2O\). But from the choices, the correct conversion factor to use here (to cancel out molecules and get moles) is \(\frac{1\space mole}{6.02\times 10^{23}\space molecules\space H_2O}\), so A is \(1\) (mole) and C is \(6.02\times 10^{23}\) (molecules \(H_2O\))? Wait, no, the question is about the units in the conversion factor. Wait, the problem is asking for the values of A and C? Wait, the original problem is a fill - in - the - blank for the conversion factor. Let's re - examine:
We have \(7.89\times 10^{24}\space molecules\space H_2O\times\frac{A\space units}{C\space units}\). To convert molecules to moles, the conversion factor is \(\frac{1\space mole}{6.02\times 10^{23}\space molecules}\). So A should be \(1\) (mole) and C should be \(6.02\times 10^{23}\) (molecules). But looking at the choices, the options are 1, \(7.89\times 10^{24}\), moles \(H_2O\), molecules \(H_2O\), \(6.02\times 10^{23}\).
So, the correct conversion factor is \(\frac{1\space mole}{6.02\times 10^{23}\space molecules\space H_2O}\), so A is \(1\) (mole) and C is \(6.02\times 10^{23}\) (molecules \(H_2O\)). But if we consider the numerical values and units:
The formula for converting molecules to moles is \(n=\frac{N}{N_A}\), where \(n\) is the number of moles, \(N\) is the number of molecules, and \(N_A = 6.02\times 10^{23}\space mol^{- 1}\).
So, \(7.89\times 10^{24}\space molecules\space H_2O\times\frac{1\space mole}{6.02\times 10^{23}\space molecules\space H_2O}\)
So, A is \(1\) (mole) and C is \(6.02\times 10^{23}\) (molecules \(H_2O\)). But from the choices, the correct values for A and C (in terms of the numbers and units) are: A is \(1\) (or \(1\space mole\)) and C is \(6.02\times 10^{23}\) (or \(6.02\times 10^{23}\space molecules\space H_2O\)). But if we look at the choices, the correct conversion factor components are: A is \(1\) (mole) and C is \(6.02\times 10^{23}\) (molecules \(H_2O\)). Wait, maybe the question is asking for the values of A and C as per the choices. Let's list the choices again: 1, \(7.89\times 10^{24}\), moles \(H_2O\), molecules \(H_2O\), \(6.02\times 10^{23}\).
So, the conversion factor is \(\frac{1\space mole}{6.02\times 10^{23}\space molecules}\), so A (numerator) should be \(1\) (mole) and C (denominator) should be \(6.02\times 10^{23}\) (molecules). But in terms of the options, the correct A is \(1\) and C is \(6.02\times 10^{23}\)? Wait, no, the units: the numerator units should be moles and the denominator units should be molecules. Wait, the problem has "A units" and "C units". So, A is \(1\) (mole) and C is \(6.02\times 10^{23}\) (molecules). But from the choic…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The value of A is \(1\) (mole) and the value of C is \(6.02\times 10^{23}\) (molecules \(H_2O\)). So in the given format, A is \(1\) and C is \(6.02\times 10^{23}\). If we consider the options, the correct A is \(1\) and C is \(6.02\times 10^{23}\).