QUESTION IMAGE
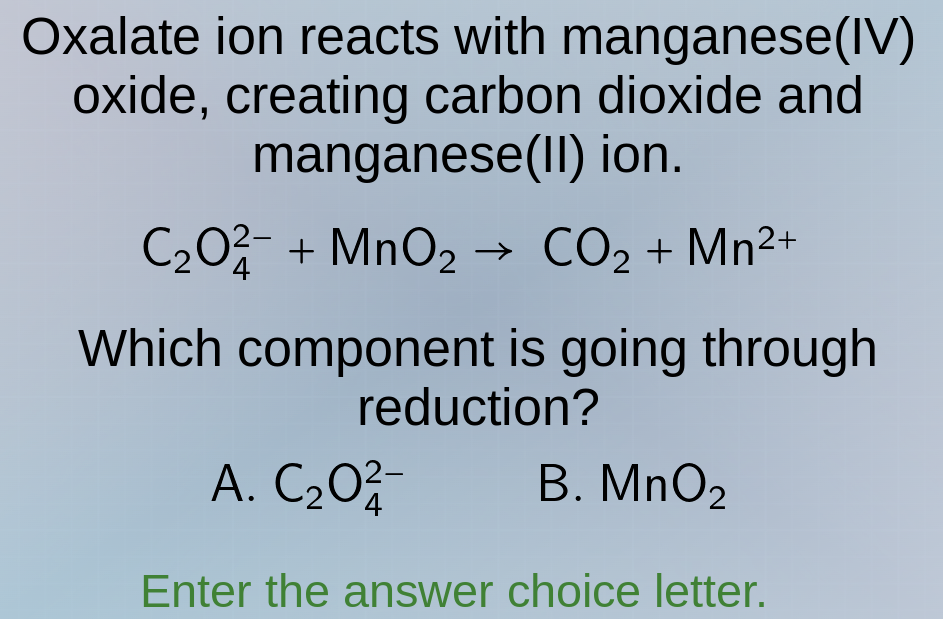
Question
oxalate ion reacts with manganese(iv) oxide, creating carbon dioxide and manganese(ii) ion.
c₂o₄²⁻ + mno₂ → co₂ + mn²⁺
which component is going through reduction?
a. c₂o₄²⁻ b. mno₂
enter the answer choice letter.
To determine which component is reduced, we analyze the oxidation states. In \(MnO_2\), Mn has an oxidation state of +4. In \(Mn^{2+}\), Mn has an oxidation state of +2. A decrease in oxidation state indicates reduction. For \(C_2O_4^{2-}\), C has an oxidation state of +3, and in \(CO_2\), C has an oxidation state of +4 (an increase, so oxidation). Thus, \(MnO_2\) (Mn's oxidation state decreases) is reduced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B