QUESTION IMAGE
Question
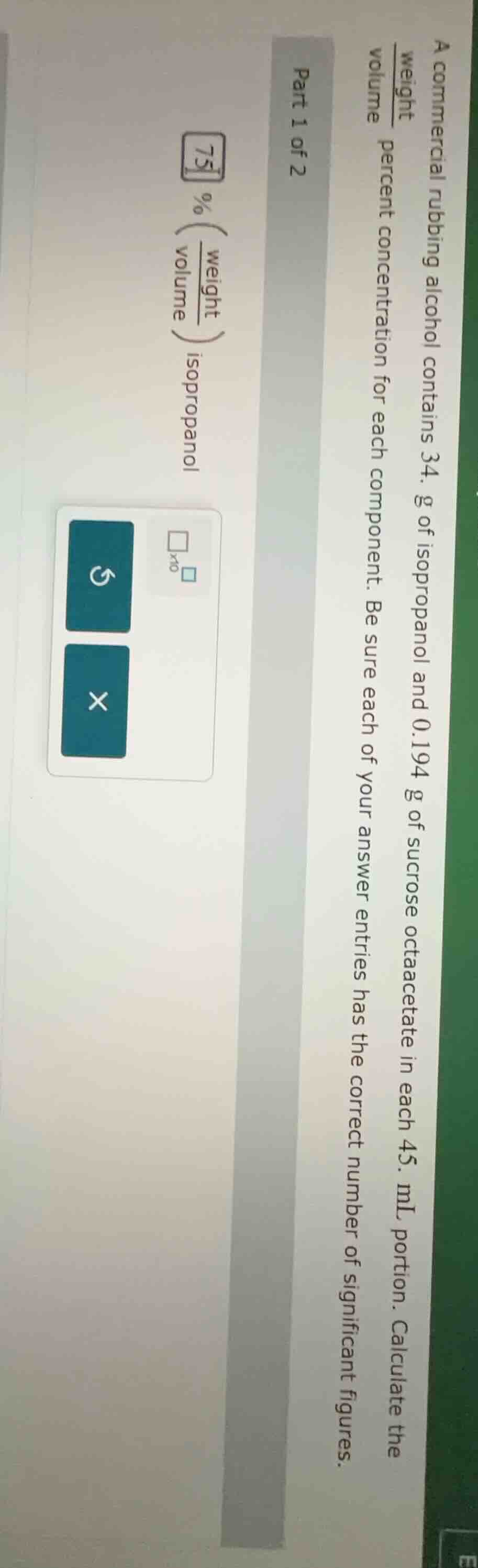
part 1 of 2
a commercial rubbing alcohol contains 34. g of isopropanol and 0.194 g of sucrose octaacetate in each 45. ml portion. calculate the $\frac{weight}{volume}$ percent concentration for each component. be sure each of your answer entries has the correct number of significant figures.
$\boldsymbol{75}$ % ($\frac{weight}{volume}$) isopropanol
Step1: Define weight/volume % formula
$\text{Weight/volume \%} = \frac{\text{mass of solute (g)}}{\text{volume of solution (mL)}} \times 100\%$
Step2: Calculate isopropanol concentration
Substitute isopropanol values: $\frac{34\ \text{g}}{45\ \text{mL}} \times 100\%$
$\approx 0.7556 \times 100\% = 76\%$
Step3: Calculate sucrose octaacetate concentration
Substitute sucrose values: $\frac{0.194\ \text{g}}{45\ \text{mL}} \times 100\%$
$\approx 0.004311 \times 100\% = 0.43\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Isopropanol: $76\%$ (weight/volume)
Sucrose octaacetate: $0.43\%$ (weight/volume)