QUESTION IMAGE
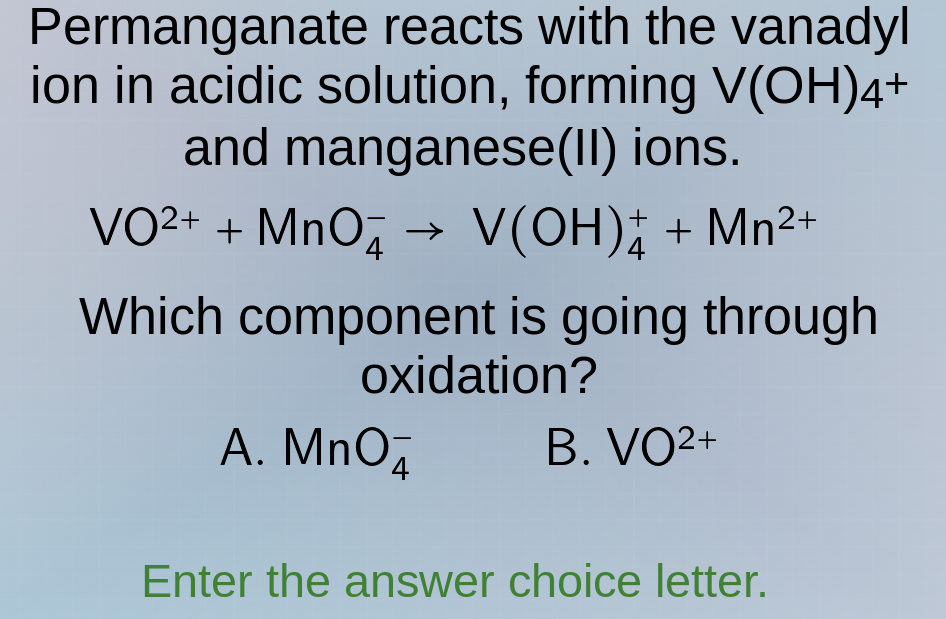
Question
permanganate reacts with the vanadyl ion in acidic solution, forming v(oh)₄⁺ and manganese(ii) ions.
vo²⁺ + mno₄⁻ → v(oh)₄⁺ + mn²⁺
which component is going through oxidation?
a. mno₄⁻ b. vo²⁺
enter the answer choice letter.
To determine oxidation, we check the change in oxidation state. For \( \text{VO}^{2+} \): In \( \text{VO}^{2+} \), let V's oxidation state be \( x \). Oxygen is -2, so \( x + (-2)= +2 \), \( x = +4 \). In \( \text{V(OH)}_4^+ \), oxygen is -2, hydrogen is +1. Let V's state be \( y \): \( y + 4(-2)+ 4(+1)= +1 \), \( y - 8 + 4 = +1 \), \( y = +5 \). V's oxidation state increases from +4 to +5, so it is oxidized. For \( \text{MnO}_4^- \): Mn in \( \text{MnO}_4^- \), let Mn's state be \( z \): \( z + 4(-2)= -1 \), \( z = +7 \). In \( \text{Mn}^{2+} \), Mn is +2, so it is reduced. Thus, \( \text{VO}^{2+} \) is oxidized.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B