QUESTION IMAGE
Question
practice: solubility curve
answer the following questions about the solubility curve below. you may need a separate sheet of paper.
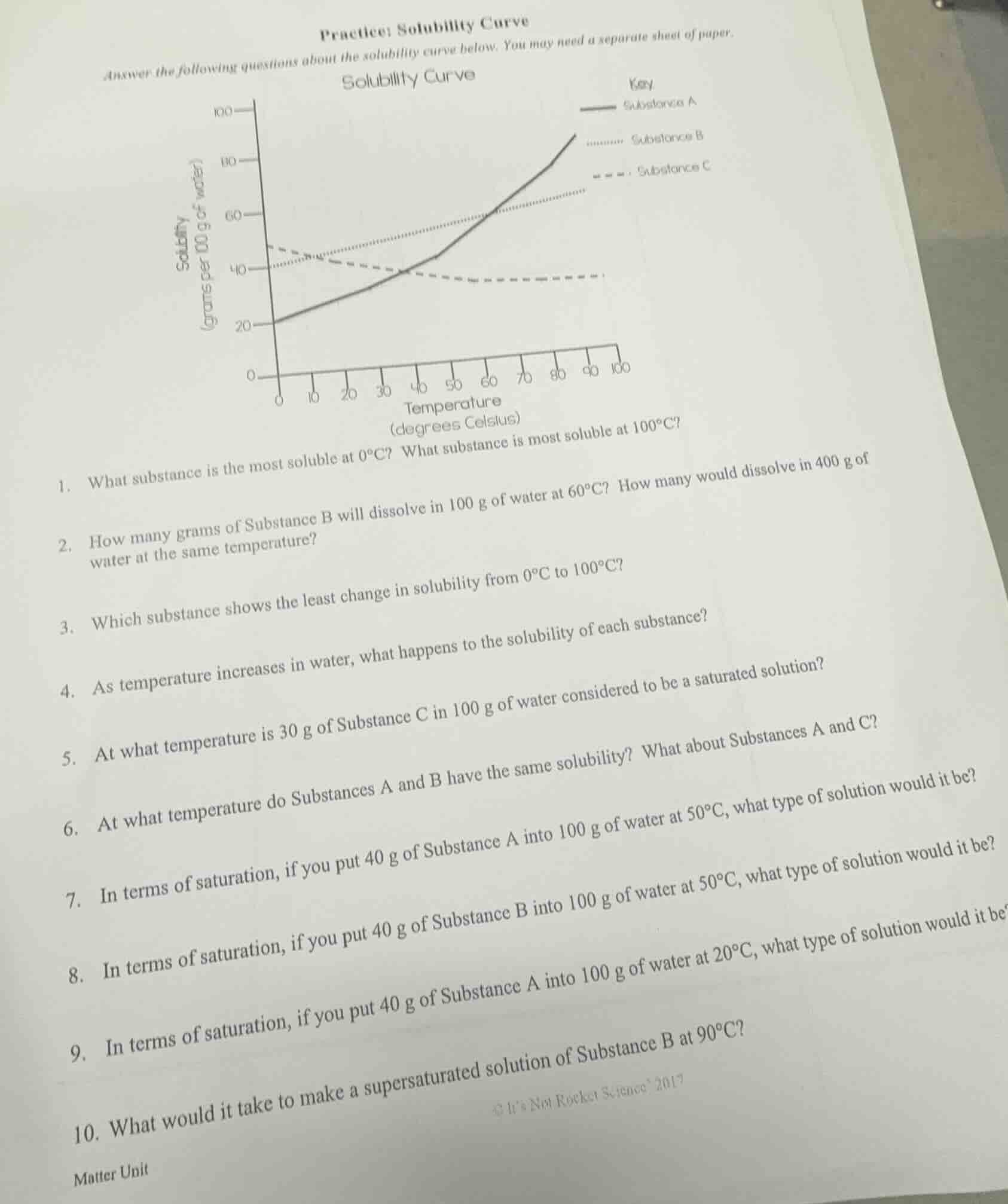
solubility curve
- what substance is the most soluble at 0°c? what substance is most soluble at 100°c?
- how many grams of substance b will dissolve in 100 g of water at 60°c? how many would dissolve in 400 g of water at the same temperature?
- which substance shows the least change in solubility from 0°c to 100°c?
- as temperature increases in water, what happens to the solubility of each substance?
- at what temperature is 30 g of substance c in 100 g of water considered to be a saturated solution?
- at what temperature do substances a and b have the same solubility? what about substances a and c?
- in terms of saturation, if you put 40 g of substance a into 100 g of water at 50°c, what type of solution would it be?
- in terms of saturation, if you put 40 g of substance b into 100 g of water at 50°c, what type of solution would it be?
- in terms of saturation, if you put 40 g of substance a into 100 g of water at 20°c, what type of solution would it be?
- what would it take to make a supersaturated solution of substance b at 90°c?
matter unit
© its not rocket science 2017
Step1: Analyze 0°C solubility
From the curve, at 0°C: Substance C (~50g) > Substance B (~40g) > Substance A (20g). At 100°C: Substance A (~90g) > Substance B (~70g) > Substance C (~35g).
Step2: Find Substance B at 60°C
At 60°C, Substance B solubility = 60g/100g water. For 400g water: $60 \times \frac{400}{100} = 240$ g.
Step3: Compare solubility change
Substance A: $90 - 20 = 70$ g; Substance B: $70 - 40 = 30$ g; Substance C: $35 - 50 = -15$ g (absolute change 15 g). Smallest change is Substance C.
Step4: Observe temperature trend
Substances A and B: solubility rises with temperature. Substance C: solubility falls with temperature.
Step5: Locate 30g Substance C
Find where Substance C hits 30g/100g water: ~50°C.
Step6: Find intersection temperatures
A and B intersect at ~60°C. A and C never intersect (curves diverge after 0°C).
Step7: Analyze Substance A at 50°C
At 50°C, Substance A solubility = 40g/100g water. Added mass = solubility: saturated.
Step8: Analyze Substance B at 50°C
At 50°C, Substance B solubility >40g/100g water. Added mass < solubility: unsaturated.
Step9: Analyze Substance A at 20°C
At 20°C, Substance A solubility = 30g/100g water. Added mass (40g) > solubility: supersaturated.
Step10: Define supersaturated solution
A supersaturated solution requires dissolving more solute than the saturation limit at 90°C, typically by heating to dissolve excess solute, then cooling slowly without disturbing the solution.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Most soluble at 0°C: Substance C; Most soluble at 100°C: Substance A
- 60 grams in 100 g water; 240 grams in 400 g water
- Substance C
- Solubility of Substance A and Substance B increases; solubility of Substance C decreases
- 50°C
- Substances A and B have the same solubility at ~60°C; Substances A and C never have the same solubility
- Saturated solution
- Unsaturated solution
- Supersaturated solution
- Dissolve more than the saturation amount of Substance B (≈70 g per 100 g water) at 90°C, typically by heating to dissolve excess solute, then cooling the solution slowly without allowing solute to crystallize out.