QUESTION IMAGE
Question
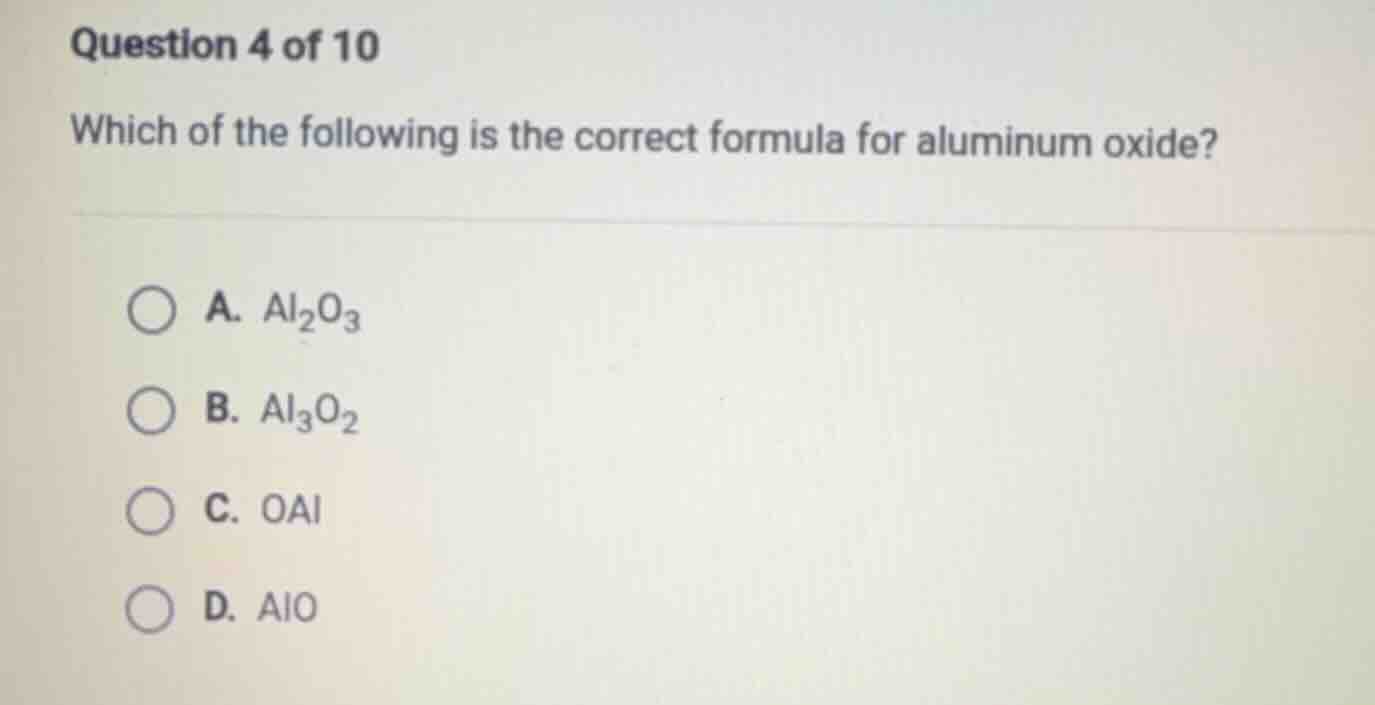
question 4 of 10
which of the following is the correct formula for aluminum oxide?
a. $\ce{al_{2}o_{3}}$
b. $\ce{al_{3}o_{2}}$
c. $\ce{oal}$
d. $\ce{alo}$
To determine the correct formula for aluminum oxide, we analyze the oxidation states of aluminum (Al) and oxygen (O). Aluminum typically has an oxidation state of +3, and oxygen typically has an oxidation state of -2. To form a neutral compound, the total positive charge must equal the total negative charge.
Let the formula be $Al_xO_y$. The charge balance gives: $x(+3) + y(-2) = 0 \implies 3x = 2y \implies \frac{x}{y} = \frac{2}{3}$. Thus, the formula is $Al_2O_3$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $Al_2O_3$