QUESTION IMAGE
Question
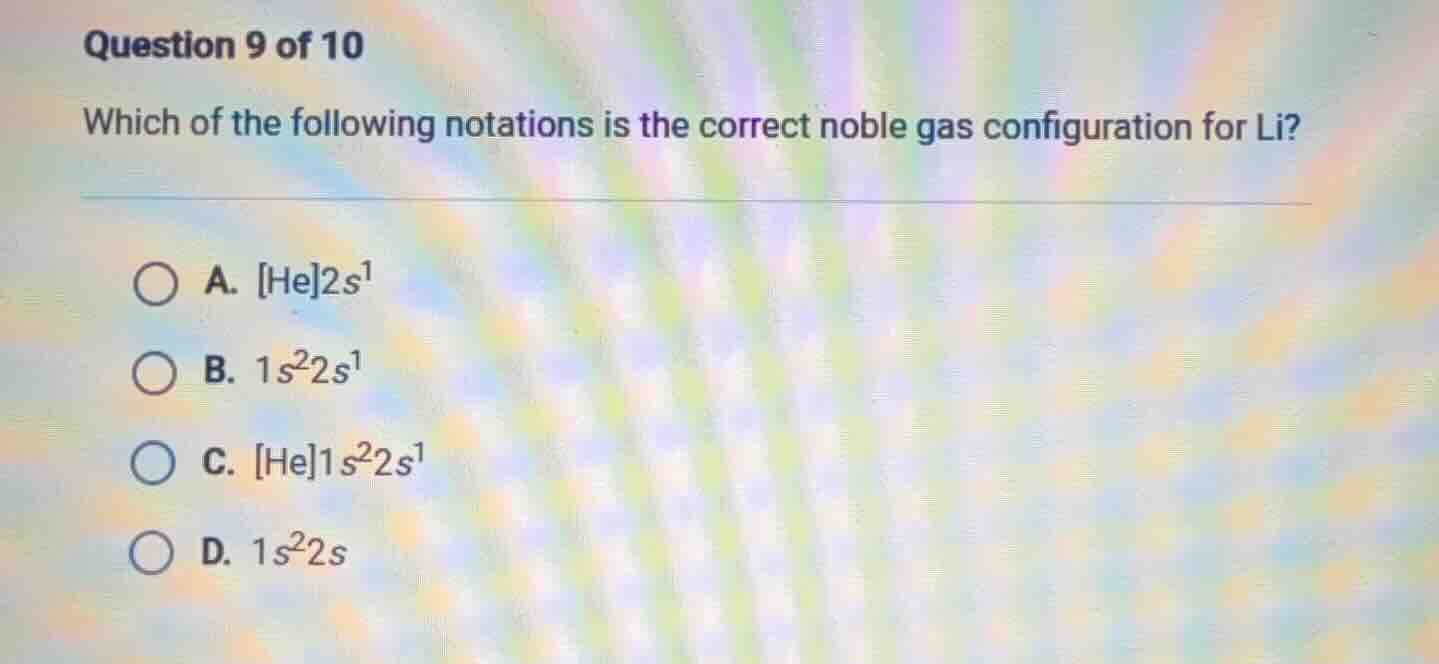
question 9 of 10
which of the following notations is the correct noble gas configuration for li?
a. he2s¹
b. 1s²2s¹
c. he1s²2s¹
d. 1s²2s
To determine the correct noble gas configuration for Lithium (Li), we first recall that noble gas configuration uses the electron configuration of the nearest noble gas (with fewer electrons than the element) and then adds the remaining electrons. The atomic number of Li is 3. The nearest noble gas with a lower atomic number is Helium (He), which has an electron configuration of $1s^2$ (or $[He]$ in noble gas notation). Lithium has 3 electrons, so after the $[He]$ (which accounts for 2 electrons), we have 1 remaining electron in the $2s$ orbital.
- Option A: $[He]2s^1$ – This uses the noble gas (He) for the first two electrons and then adds the remaining electron in $2s^1$, which is correct for noble gas configuration.
- Option B: $1s^22s^1$ – This is the full electron configuration, not the noble gas configuration.
- Option C: $[He]1s^22s^1$ – This is incorrect because $[He]$ already represents $1s^2$, so repeating $1s^2$ is redundant.
- Option D: $1s^22s$ – This is an incomplete electron configuration (missing the exponent on $2s$) and not a noble gas configuration.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $[He]2s^1$