QUESTION IMAGE
Question
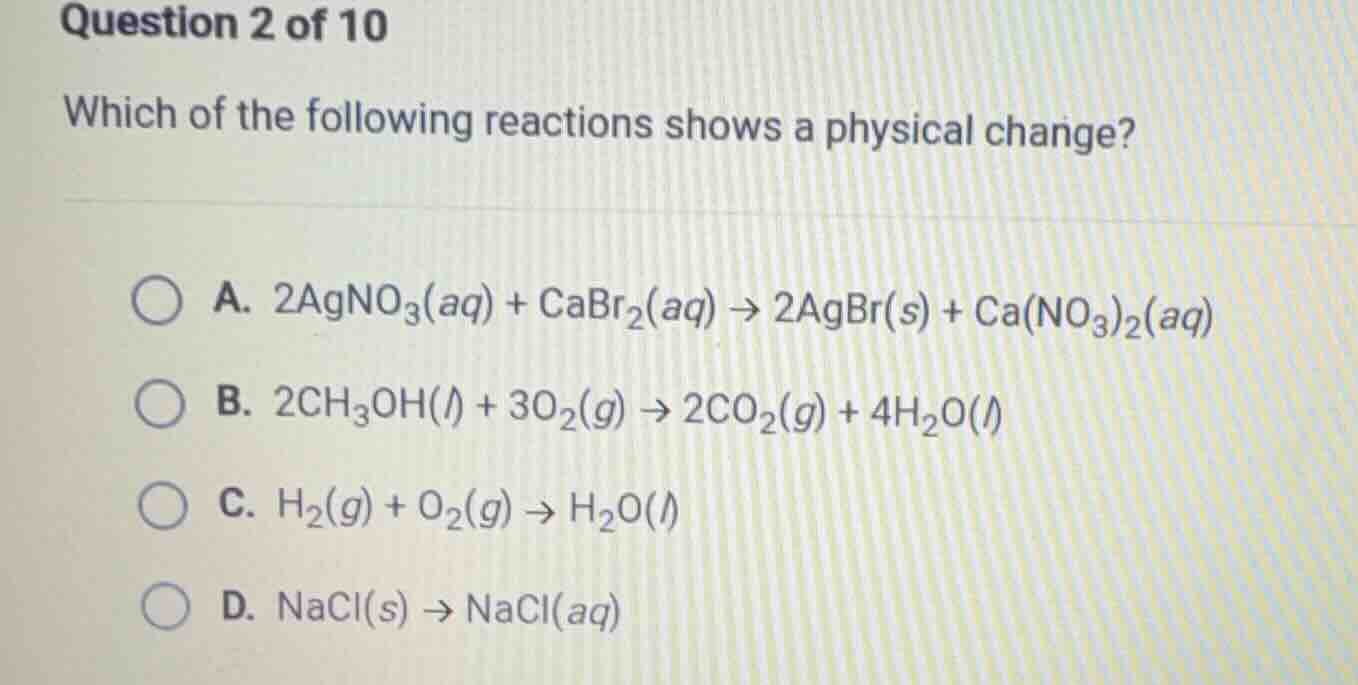
question 2 of 10
which of the following reactions shows a physical change?
a. $2\ce{agno3}(aq) + \ce{cabr2}(aq) \
ightarrow 2\ce{agbr}(s) + \ce{ca(no3)2}(aq)$
b. $2\ce{ch3oh}(l) + 3\ce{o2}(g) \
ightarrow 2\ce{co2}(g) + 4\ce{h2o}(l)$
c. $\ce{h2}(g) + \ce{o2}(g) \
ightarrow \ce{h2o}(l)$
d. $\ce{nacl}(s) \
ightarrow \ce{nacl}(aq)$
To determine a physical change, we check if new substances are formed. A physical change doesn't create new chemical substances, just changes state or form.
- Option A: A precipitation reaction, new solid (AgBr) and aqueous compound form—chemical change.
- Option B: Combustion of methanol, new substances (CO₂, H₂O) formed—chemical change.
- Option C: Formation of water from hydrogen and oxygen, new substance (H₂O) formed—chemical change.
- Option D: NaCl(s) dissolving to NaCl(aq) is dissolving (solvation), no new substance (still NaCl, just dispersed in water)—physical change.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\text{NaCl}(s)
ightarrow \text{NaCl}(aq)$