QUESTION IMAGE
Question
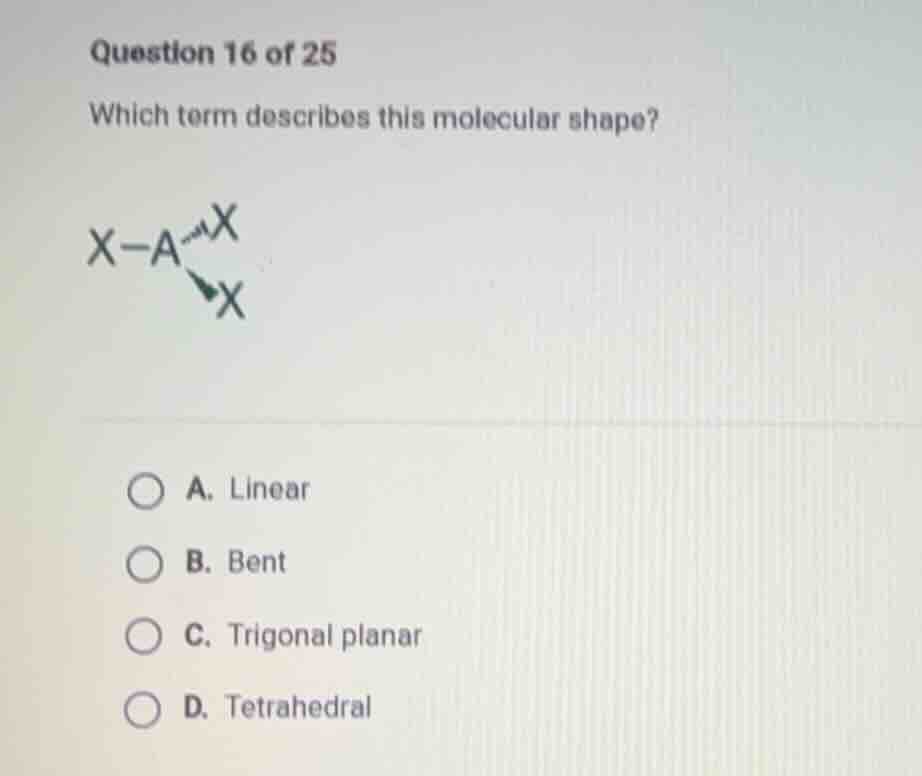
question 16 of 25
which term describes this molecular shape?
x - a - x
\\ /
x
a. linear
b. bent
c. trigonal planar
d. tetrahedral
To determine the molecular shape, we analyze the structure: the central atom \( A \) is bonded to three \( X \) atoms, and the arrangement (with three bonding regions) corresponds to trigonal planar geometry. Linear has two bonding regions, bent has a non - linear arrangement with lone pairs (not indicated here), and tetrahedral has four regions. So the shape is trigonal planar.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Trigonal planar