QUESTION IMAGE
Question
question 32 of 34
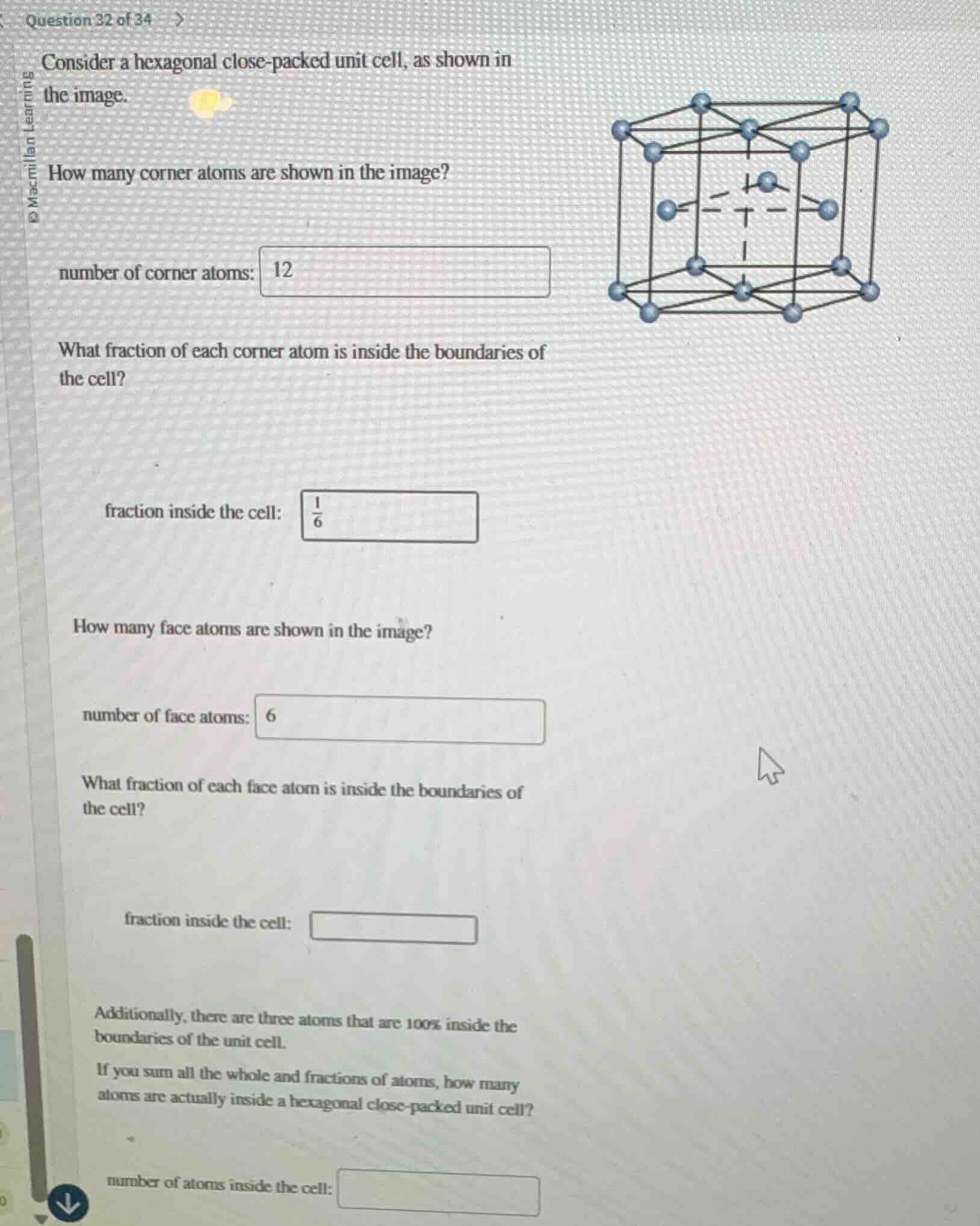
consider a hexagonal close - packed unit cell, as shown in
the image.
how many corner atoms are shown in the image?
number of corner atoms: 12
what fraction of each corner atom is inside the boundaries of
the cell?
fraction inside the cell: $\frac{1}{6}$
how many face atoms are shown in the image?
number of face atoms: 6
what fraction of each face atom is inside the boundaries of
the cell?
fraction inside the cell:
additionally, there are three atoms that are 100% inside the
boundaries of the unit cell.
if you sum all the whole and fractions of atoms, how many
atoms are actually inside a hexagonal close - packed unit cell?
number of atoms inside the cell:
Step1: Face atom fraction definition
Face atoms are shared equally between 2 adjacent unit cells, so the fraction inside one cell is $\frac{1}{2}$.
Step2: Calculate corner atom contribution
Multiply corner count by their fraction:
$12 \times \frac{1}{6} = 2$
Step3: Calculate face atom contribution
Multiply face count by their fraction:
$6 \times \frac{1}{2} = 3$
Step4: Add internal atoms
Add the 3 fully internal atoms to the contributions:
$2 + 3 + 3 = 6$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
fraction inside the cell: $\frac{1}{2}$
number of atoms inside the cell: 6