QUESTION IMAGE
Question
question 35 of 60
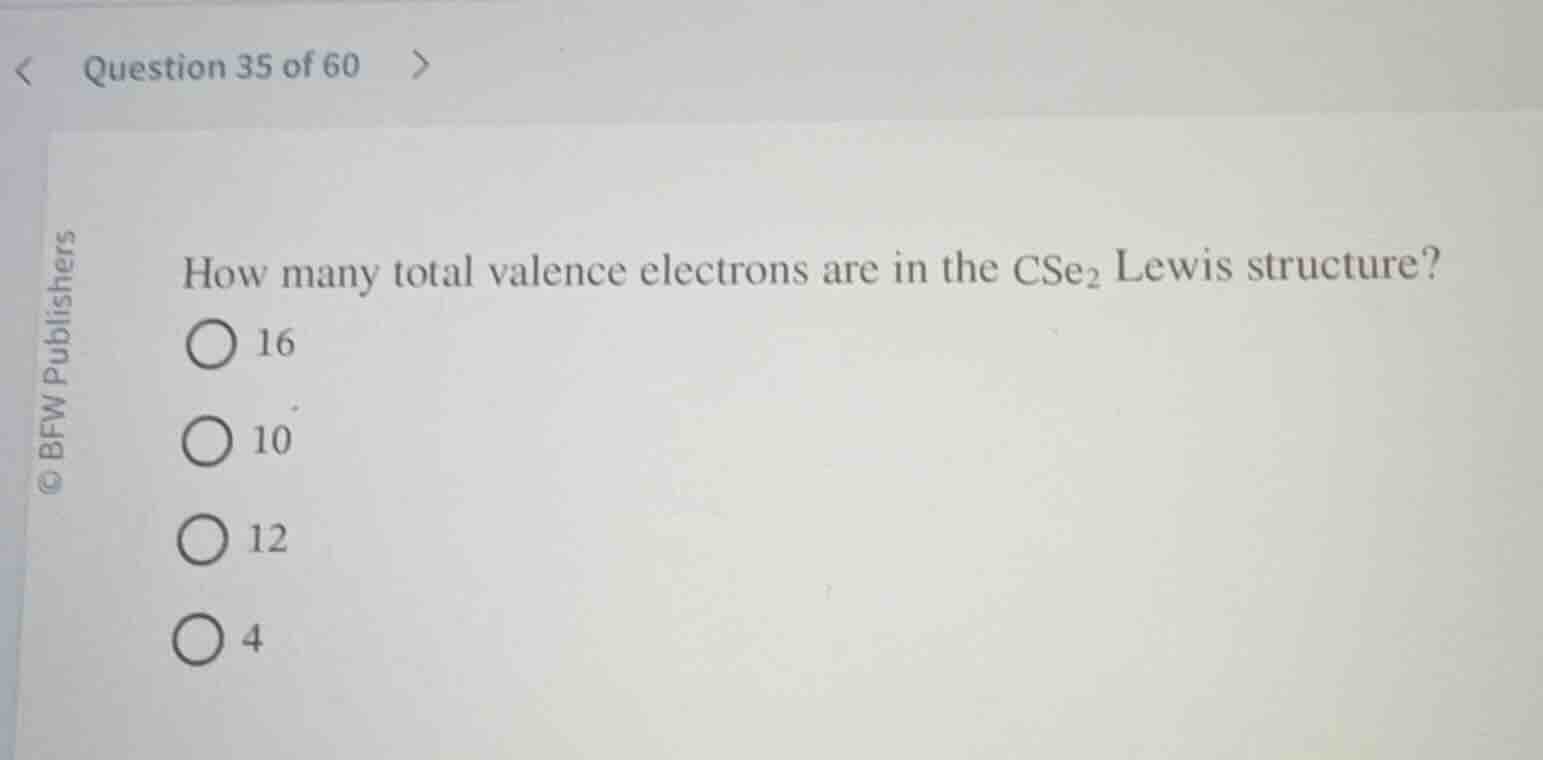
how many total valence electrons are in the $\text{cse}_2$ lewis structure?
$\bigcirc$ 16
$\bigcirc$ 10
$\bigcirc$ 12
$\bigcirc$ 4
© bfw publishers
Step1: Find valence electrons of each atom
Carbon (C): 4 valence electrons; Selenium (Se): 6 valence electrons each.
Step2: Calculate total valence electrons
Total = Valence electrons of C + 2×Valence electrons of Se
$\text{Total} = 4 + 2\times6 = 4 + 12 = 16$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
16