QUESTION IMAGE
Question
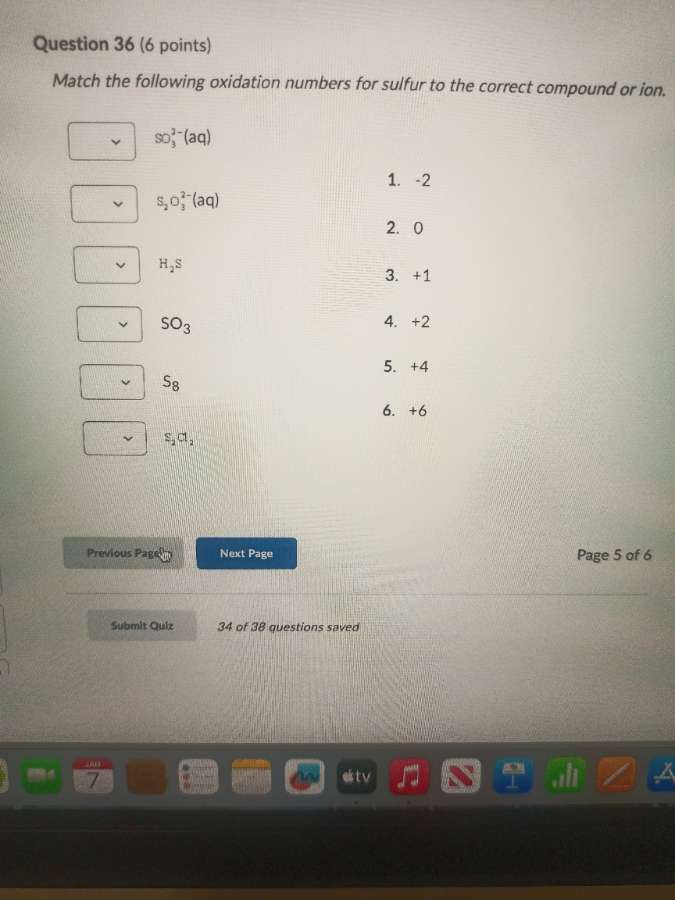
question 36 (6 points)
match the following oxidation numbers for sulfur to the correct compound or ion.
\\(\ce{so^2-_{3}(aq)}\\)
\\(\ce{s_{2}o^2-_{3}(aq)}\\)
\\(\ce{h_{2}s}\\)
\\(\ce{so_{3}}\\)
\\(\ce{s_{8}}\\)
\\(\ce{s_{2}cl_{2}}\\)
- -2
- 0
- +1
- +2
- +4
- +6
previous page next page
page 5 of 6
submit quiz 34 of 38 questions saved
To determine the oxidation number of sulfur in each compound/ion, we use the rules for oxidation numbers (e.g., oxygen is usually -2, hydrogen is +1, the sum of oxidation numbers in a neutral compound is 0, in a polyatomic ion is equal to the ion's charge).
Step 1: $\boldsymbol{SO_3^{2-}}$ (aq)
Let the oxidation number of S be $x$. Oxygen is -2, and the ion has a charge of -2.
Sum of oxidation numbers: $x + 3(-2) = -2$
$x - 6 = -2$
$x = +4$
Step 2: $\boldsymbol{S_2O_3^{2-}}$ (aq)
Let S be $x$. Oxygen is -2, charge is -2.
$2x + 3(-2) = -2$
$2x - 6 = -2$
$2x = 4$
$x = +2$
Step 3: $\boldsymbol{H_2S}$
Hydrogen is +1. Let S be $x$. Neutral compound, so sum is 0.
$2(+1) + x = 0$
$x = -2$
Step 4: $\boldsymbol{SO_3}$
Oxygen is -2. Let S be $x$. Neutral compound.
$x + 3(-2) = 0$
$x - 6 = 0$
$x = +6$
Step 5: $\boldsymbol{S_8}$
Elemental sulfur (molecule of S atoms), so oxidation number is 0.
Step 6: $\boldsymbol{S_2Cl_2}$
Chlorine is -1. Let S be $x$. Neutral compound.
$2x + 2(-1) = 0$
$2x - 2 = 0$
$2x = 2$
$x = +1$
Matching:
- $SO_3^{2-}$ (aq) → +4 (Option 5)
- $S_2O_3^{2-}$ (aq) → +2 (Option 4)
- $H_2S$ → -2 (Option 1)
- $SO_3$ → +6 (Option 6)
- $S_8$ → 0 (Option 2)
- $S_2Cl_2$ → +1 (Option 3)
(Note: Present the final matches as per the question's dropdowns, but since the question asks to "match" and the solution shows the oxidation numbers, the above steps confirm the correct pairings.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the oxidation number of sulfur in each compound/ion, we use the rules for oxidation numbers (e.g., oxygen is usually -2, hydrogen is +1, the sum of oxidation numbers in a neutral compound is 0, in a polyatomic ion is equal to the ion's charge).
Step 1: $\boldsymbol{SO_3^{2-}}$ (aq)
Let the oxidation number of S be $x$. Oxygen is -2, and the ion has a charge of -2.
Sum of oxidation numbers: $x + 3(-2) = -2$
$x - 6 = -2$
$x = +4$
Step 2: $\boldsymbol{S_2O_3^{2-}}$ (aq)
Let S be $x$. Oxygen is -2, charge is -2.
$2x + 3(-2) = -2$
$2x - 6 = -2$
$2x = 4$
$x = +2$
Step 3: $\boldsymbol{H_2S}$
Hydrogen is +1. Let S be $x$. Neutral compound, so sum is 0.
$2(+1) + x = 0$
$x = -2$
Step 4: $\boldsymbol{SO_3}$
Oxygen is -2. Let S be $x$. Neutral compound.
$x + 3(-2) = 0$
$x - 6 = 0$
$x = +6$
Step 5: $\boldsymbol{S_8}$
Elemental sulfur (molecule of S atoms), so oxidation number is 0.
Step 6: $\boldsymbol{S_2Cl_2}$
Chlorine is -1. Let S be $x$. Neutral compound.
$2x + 2(-1) = 0$
$2x - 2 = 0$
$2x = 2$
$x = +1$
Matching:
- $SO_3^{2-}$ (aq) → +4 (Option 5)
- $S_2O_3^{2-}$ (aq) → +2 (Option 4)
- $H_2S$ → -2 (Option 1)
- $SO_3$ → +6 (Option 6)
- $S_8$ → 0 (Option 2)
- $S_2Cl_2$ → +1 (Option 3)
(Note: Present the final matches as per the question's dropdowns, but since the question asks to "match" and the solution shows the oxidation numbers, the above steps confirm the correct pairings.)