QUESTION IMAGE
Question
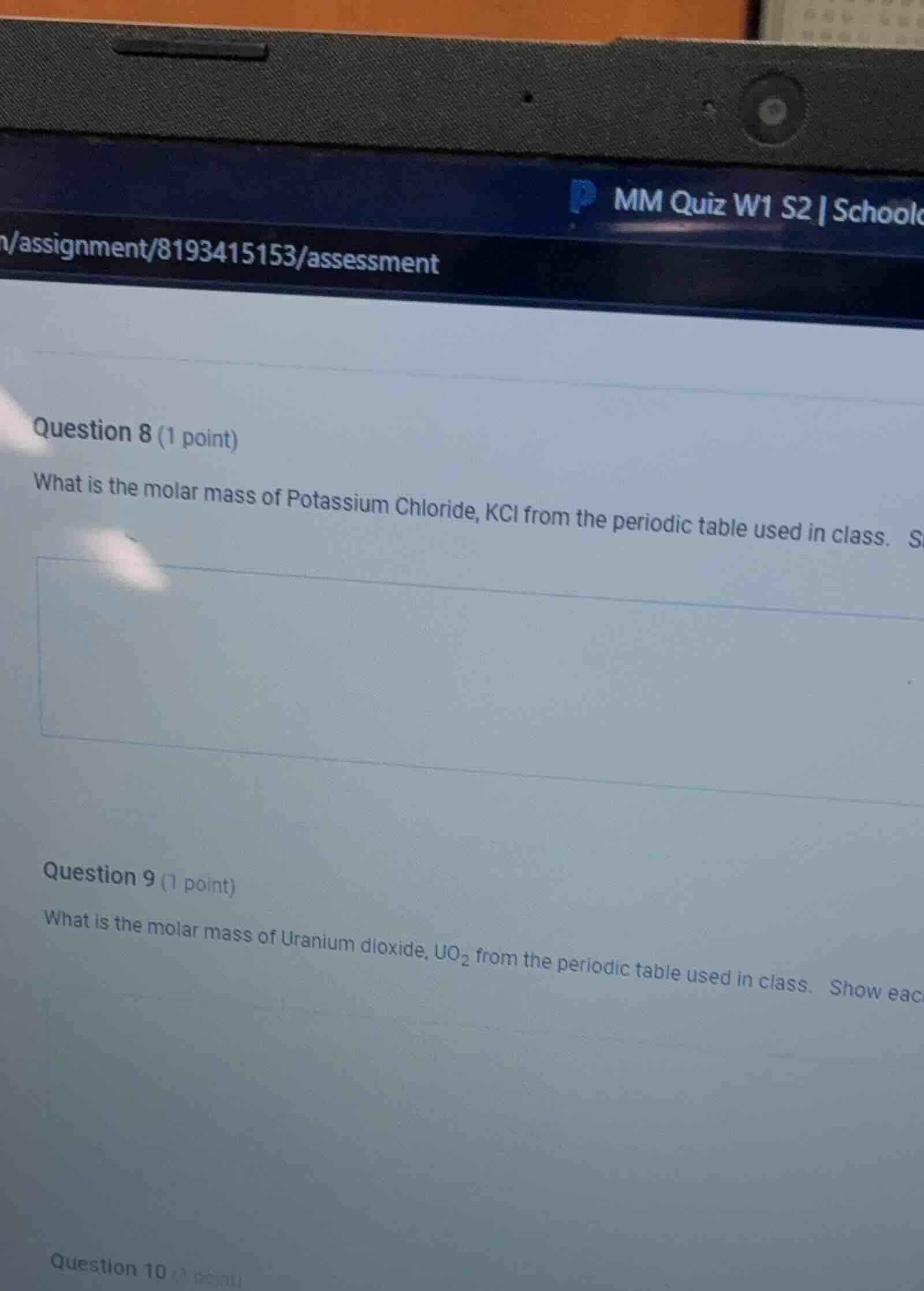
question 8 (1 point)
what is the molar mass of potassium chloride, kcl from the periodic table used in class.
question 9 (1 point)
what is the molar mass of uranium dioxide, uo₂ from the periodic table used in class. show eac
Response
Question 8:
Step1: Find atomic masses
Potassium (K) atomic mass ≈ 39.10 g/mol, Chlorine (Cl) atomic mass ≈ 35.45 g/mol.
Step2: Sum atomic masses
Molar mass of KCl = 39.10 + 35.45 = 74.55 g/mol.
Step1: Find atomic masses
Uranium (U) atomic mass ≈ 238.03 g/mol, Oxygen (O) atomic mass ≈ 16.00 g/mol.
Step2: Calculate for \( UO_2 \)
Molar mass = 238.03 + (2 × 16.00) = 238.03 + 32.00 = 270.03 g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
74.55 g/mol