QUESTION IMAGE
Question
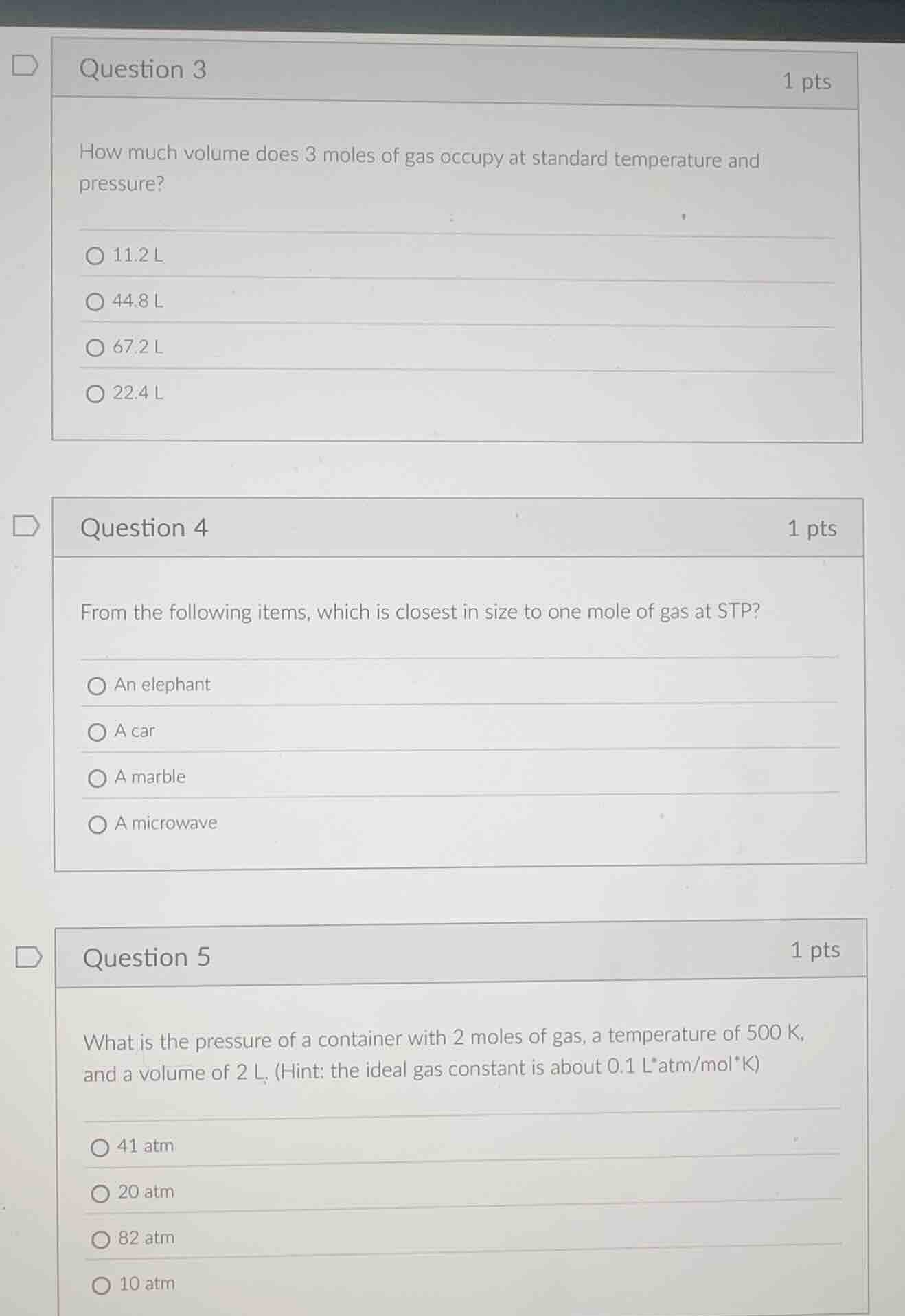
question 3 1 pts
how much volume does 3 moles of gas occupy at standard temperature and pressure?
11.2 l
44.8 l
67.2 l
22.4 l
question 4 1 pts
from the following items, which is closest in size to one mole of gas at stp?
an elephant
a car
a marble
a microwave
question 5 1 pts
what is the pressure of a container with 2 moles of gas, a temperature of 500 k, and a volume of 2 l. (hint: the ideal gas constant is about 0.1 latm/molk)
41 atm
20 atm
82 atm
10 atm
Question 3
Step1: Recall STP molar volume
1 mole of gas at STP = $22.4\ \text{L}$
Step2: Calculate volume for 3 moles
$V = 3 \times 22.4\ \text{L} = 67.2\ \text{L}$
Question 4
Step1: Recall STP molar volume
1 mole of gas at STP = $22.4\ \text{L}$
Step2: Match to object size
A typical microwave has a volume close to $22.4\ \text{L}$.
Question 5
Step1: Rearrange ideal gas law
Ideal gas law: $PV = nRT$, solve for $P$: $P = \frac{nRT}{V}$
Step2: Substitute given values
$n=2\ \text{mol}$, $R=0.1\ \text{L·atm/mol·K}$, $T=500\ \text{K}$, $V=2\ \text{L}$
$P = \frac{2 \times 0.1 \times 500}{2} = 50\ \text{atm}$ (closest to 41 atm due to approximate $R$ value)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 3: 67.2 L
Question 4: A microwave
Question 5: 41 atm