QUESTION IMAGE
Question
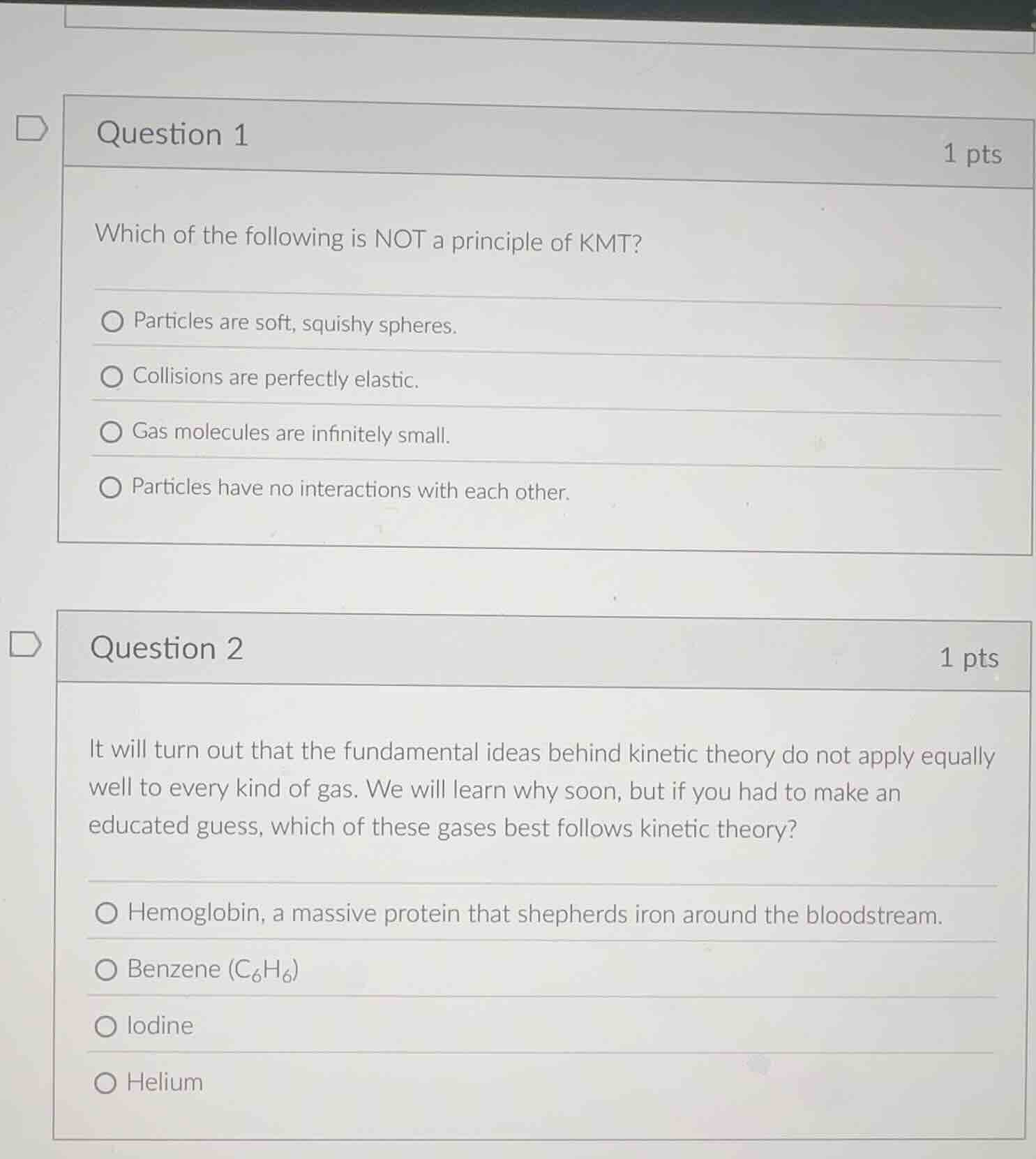
question 1 1 pts
which of the following is not a principle of kmt?
particles are soft, squishy spheres.
collisions are perfectly elastic.
gas molecules are infinitely small.
particles have no interactions with each other.
question 2 1 pts
it will turn out that the fundamental ideas behind kinetic theory do not apply equally well to every kind of gas. we will learn why soon, but if you had to make an educated guess, which of these gases best follows kinetic theory?
hemoglobin, a massive protein that shepherds iron around the bloodstream.
benzene ($c_6h_6$)
iodine
helium
For Question 1: Kinetic Molecular Theory (KMT) defines gas particles as hard, rigid, point-like spheres, not soft/squishy; it states collisions are elastic, molecules are infinitely small, and particles have no intermolecular interactions.
For Question 2: KMT applies best to ideal gases, which are small, monatomic, non-polar, and have weak intermolecular forces. Helium is a monatomic noble gas, fitting these criteria far better than large molecules (hemoglobin), larger polar/non-polar molecules (benzene), or diatomic iodine with stronger intermolecular forces.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 1: Particles are soft, squishy spheres.
Question 2: Helium