QUESTION IMAGE
Question
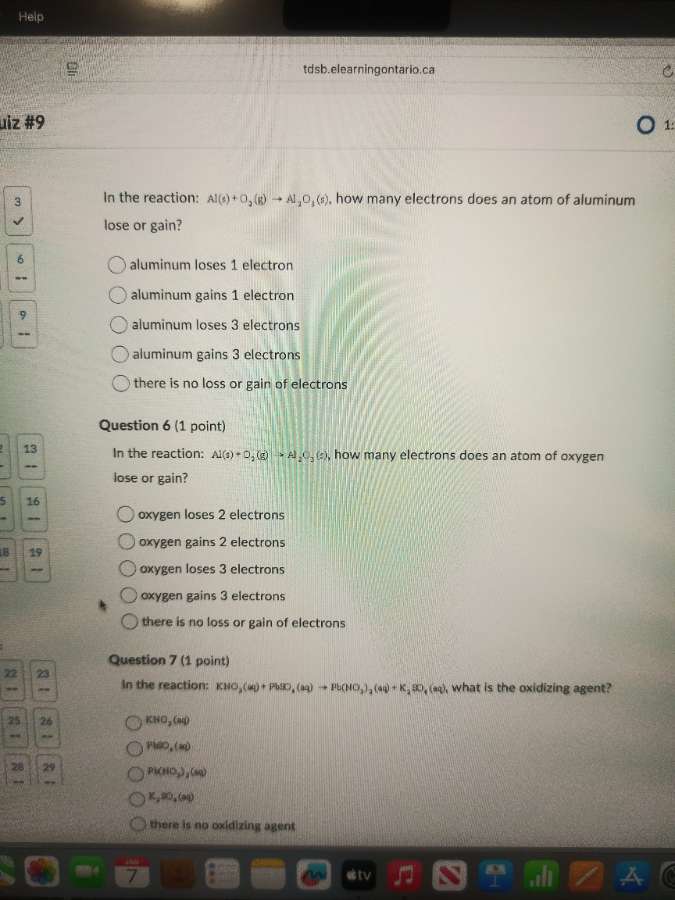
in the reaction: al(s) + o₂(g) → al₂o₃(s), how many electrons does an atom of aluminum lose or gain?
○ aluminum loses 1 electron
○ aluminum gains 1 electron
○ aluminum loses 3 electrons
○ aluminum gains 3 electrons
○ there is no loss or gain of electrons
question 6 (1 point)
in the reaction: al(s) + o₂(g) → al₂o₃(s), how many electrons does an atom of oxygen lose or gain?
○ oxygen loses 2 electrons
○ oxygen gains 2 electrons
○ oxygen loses 3 electrons
○ oxygen gains 3 electrons
○ there is no loss or gain of electrons
question 7 (1 point)
in the reaction: kno₃(aq) + pbs(s) → pb(no₃)₂(aq) + k₂s(aq), what is the oxidizing agent?
○ kno₃(aq)
○ pbs(s)
○ pb(no₃)₂(aq)
○ k₂s(aq)
○ there is no oxidizing agent
Question (Aluminum electron change)
In the reaction \( \text{Al}(s) + \text{O}_2(g)
ightarrow \text{Al}_2\text{O}_3(s) \), aluminum (Al) starts with an oxidation state of 0 (in elemental form). In \( \text{Al}_2\text{O}_3 \), oxygen has an oxidation state of -2. Let the oxidation state of Al be \( x \). For \( \text{Al}_2\text{O}_3 \), we have \( 2x + 3(-2) = 0 \), so \( 2x - 6 = 0 \), and \( x = +3 \). So Al goes from 0 to +3, meaning it loses 3 electrons (since oxidation is loss of electrons, and an increase in oxidation state indicates oxidation).
In \( \text{Al}(s) + \text{O}_2(g)
ightarrow \text{Al}_2\text{O}_3(s) \), oxygen (O) in \( \text{O}_2 \) (elemental form) has an oxidation state of 0. In \( \text{Al}_2\text{O}_3 \), oxygen has an oxidation state of -2. A decrease in oxidation state (from 0 to -2) means oxygen gains electrons (reduction is gain of electrons). The change per O atom is \( 0 - (-2) = 2 \), so each O atom gains 2 electrons.
In the reaction \( \text{KNO}_3(aq) + \text{PbS}(aq)
ightarrow \text{Pb(NO}_3\text{)}_2(aq) + \text{K}_2\text{S}(aq) \), we check oxidation states. For \( \text{KNO}_3 \): K is +1, N is +5, O is -2. For \( \text{PbS} \): Pb is +2, S is -2. In products, \( \text{Pb(NO}_3\text{)}_2 \): Pb is +2, N is +5, O is -2; \( \text{K}_2\text{S} \): K is +1, S is -2. Wait, maybe there's a typo or I misread? Wait, no—wait, maybe the reaction is different? Wait, no, in the given reaction, let's check oxidation states. Wait, actually, maybe the reaction is a double - displacement? But double - displacement reactions (like this, swapping ions: \( \text{K}^+ \) with \( \text{Pb}^{2+} \), \( \text{NO}_3^- \) with \( \text{S}^{2-} \)) are not redox reactions (no change in oxidation states). But that can't be. Wait, maybe the reaction is written wrong? Wait, no, the options include \( \text{KNO}_3(aq) \) as a candidate. Wait, no—if it's a double - displacement, there's no oxidizing agent. But that seems odd. Wait, maybe the reaction is supposed to be a redox? Wait, no, in \( \text{KNO}_3(aq) + \text{PbS}(aq)
ightarrow \text{Pb(NO}_3\text{)}_2(aq) + \text{K}_2\text{S}(aq) \), the oxidation states: K is +1 (reactant and product), N is +5 (reactant and product), O is -2, Pb is +2 (reactant and product), S is -2 (reactant and product). So no change in oxidation states, meaning it's not a redox reaction, so there is no oxidizing agent. But wait, maybe the reaction was mistyped. But based on the given reaction, since there's no change in oxidation states (all oxidation states remain the same), there is no oxidizing agent. Wait, but the options have \( \text{KNO}_3(aq) \), \( \text{PbS}(aq) \), etc. Wait, maybe I made a mistake. Wait, no—double - displacement reactions (ion exchange) are not redox. So in this reaction, since it's a double - displacement (forming \( \text{Pb(NO}_3\text{)}_2 \) and \( \text{K}_2\text{S} \) from \( \text{KNO}_3 \) and \( \text{PbS} \)), there's no oxidation or reduction, so no oxidizing agent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
aluminum loses 3 electrons