QUESTION IMAGE
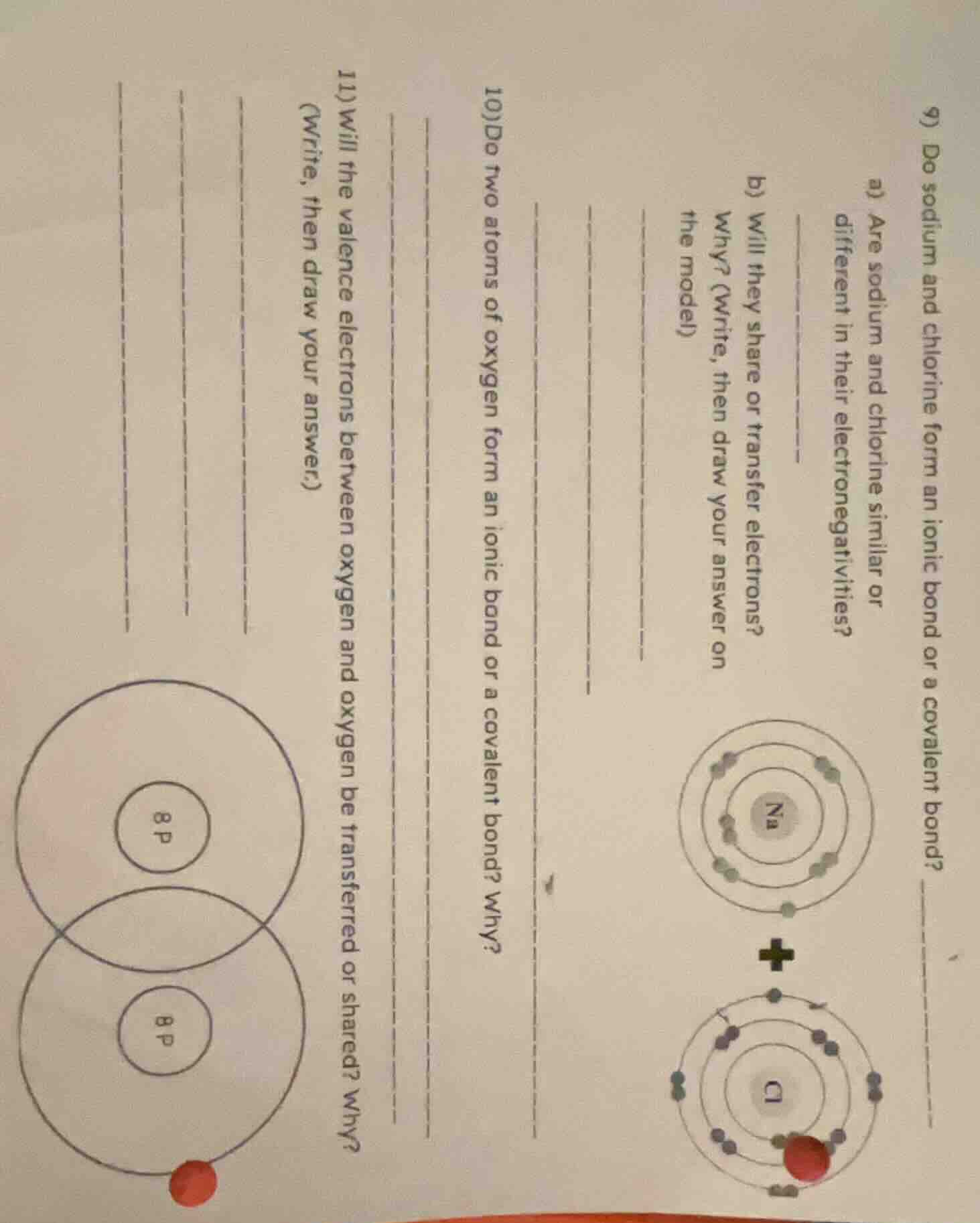
Question
- do sodium and chlorine form an ionic bond or a covalent bond?
a) are sodium and chlorine similar or different in their electronegativities?
b) will they share or transfer electrons? why? (write, then draw your answer on the model)
- do two atoms of oxygen form an ionic bond or a covalent bond? why?
- will the valence electrons between oxygen and oxygen be transferred or shared? why? (write, then draw your answer.)
Problem 9
a)
Sodium (metal) y cloro (no metal) tienen electronegatividades diferentes (Na es electropositivo, Cl es electronegativo). La diferencia de electronegatividad es grande.
Sodium (Na) tiene 1 electrón de valencia, Cl tiene 7. Na transfiere su electrón de valencia a Cl (Na⁺, Cl⁻), formando enlace iónico (transferencia, no compartición).
Dos átomos de oxígeno (no metales, misma electronegatividad) comparten electrones para completar octetos (enlace covalente).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Sodium y chlorine son diferentes en sus electronegatividades.