QUESTION IMAGE
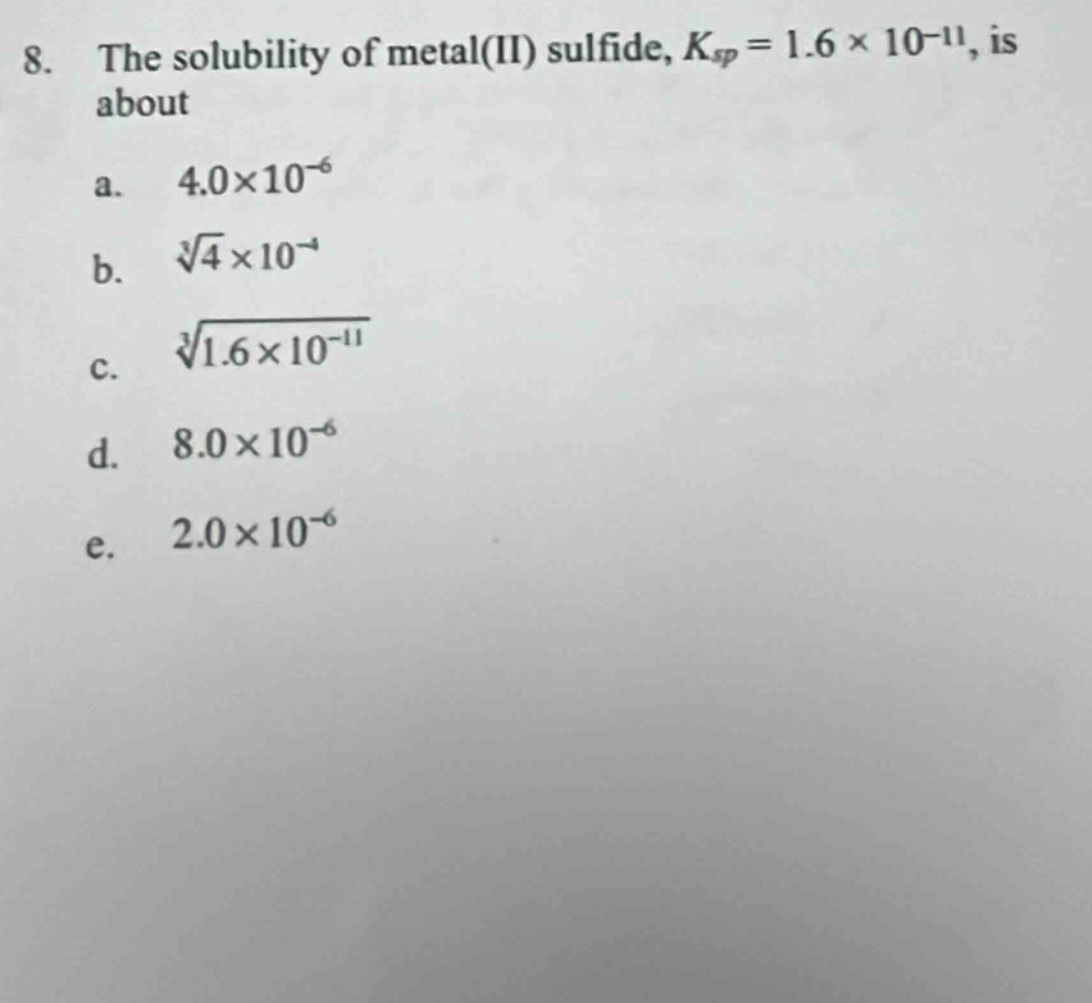
Question
- the solubility of metal(ii) sulfide, $k_{sp}=1.6 \times 10^{-11}$, is about
a. $4.0\times10^{-6}$
b. $\sqrt3{4}\times10^{-4}$
c. $\sqrt3{1.6\times10^{-11}}$
d. $8.0\times10^{-6}$
e. $2.0\times10^{-6}$
Step1: Write dissolution equation
Metal(II) sulfide (let's say \( MS \)) dissolves as: \( MS(s)
ightleftharpoons M^{2+}(aq) + S^{2-}(aq) \). Let solubility be \( s \), so \([M^{2+}] = s\), \([S^{2-}] = s\).
Step2: Write \( K_{sp} \) expression
\( K_{sp} = [M^{2+}][S^{2-}] = s \times s = s^2 \)? Wait, no, wait—wait, metal(II) sulfide: wait, no, maybe I made a mistake. Wait, metal(II) sulfide: formula is \( MS \), so dissociation is \( MS
ightarrow M^{2+} + S^{2-} \), so \( K_{sp} = [M^{2+}][S^{2-}] = s \times s = s^2 \)? But the given \( K_{sp} = 1.6 \times 10^{-11} \), so \( s = \sqrt{1.6 \times 10^{-11}} \)? Wait, no, that can't be. Wait, maybe I misread the formula. Wait, maybe it's a metal(II) sulfide with a different formula? Wait, no, metal(II) sulfide is \( MS \), so 1:1 ratio. Wait, but the options have cube roots. Wait, maybe the formula is \( M_2S \)? No, metal(II) sulfide: metal is +2, sulfide is -2, so \( MS \). Wait, maybe the problem is a typo, or maybe I made a mistake. Wait, let's check the options. Option b is \( \sqrt[3]{4} \times 10^{-4} \), option c is \( \sqrt[3]{1.6 \times 10^{-11}} \). Wait, maybe the formula is \( M_2S \)? No, metal(II) sulfide: \( MS \). Wait, maybe the solubility is for a compound with formula \( M_2S_3 \)? No, the problem says metal(II) sulfide, so \( MS \). Wait, maybe I miscalculated. Wait, \( K_{sp} = 1.6 \times 10^{-11} \). If \( MS \) dissociates into \( M^{2+} \) and \( S^{2-} \), then \( K_{sp} = s^2 \), so \( s = \sqrt{1.6 \times 10^{-11}} \). Let's calculate that: \( 1.6 \times 10^{-11} = 16 \times 10^{-12} \), so \( \sqrt{16 \times 10^{-12}} = 4 \times 10^{-6} \). Wait, option a is \( 4.0 \times 10^{-6} \). But wait, the cube roots in options b and c suggest a 1:2 or 2:1 ratio. Wait, maybe the formula is \( M_2S \) (metal(I) sulfide) or \( MS_2 \) (metal(IV) sulfide). Wait, metal(II) sulfide: \( MS \), so 1:1. But let's recalculate \( \sqrt{1.6 \times 10^{-11}} \). \( 1.6 \times 10^{-11} = 16 \times 10^{-12} \), square root of 16 is 4, square root of \( 10^{-12} \) is \( 10^{-6} \), so \( \sqrt{16 \times 10^{-12}} = 4 \times 10^{-6} \), which is option a. But wait, the cube roots in options b and c. Maybe the problem is actually a metal(III) sulfide? No, the problem says metal(II) sulfide. Wait, maybe the original problem was metal(III) sulfide, \( M_2S_3 \), which dissociates into \( 2M^{3+} + 3S^{2-} \), so \( K_{sp} = (2s)^2(3s)^3 = 4s^2 \times 27s^3 = 108s^5 \), but that's not matching. Wait, no, let's check the given \( K_{sp} = 1.6 \times 10^{-11} \). If we assume the formula is \( MS \), then \( K_{sp} = s^2 \), so \( s = \sqrt{1.6 \times 10^{-11}} = \sqrt{16 \times 10^{-12}} = 4 \times 10^{-6} \), which is option a. But let's check option a: \( 4.0 \times 10^{-6} \). Let's square that: \( (4 \times 10^{-6})^2 = 16 \times 10^{-12} = 1.6 \times 10^{-11} \), which matches \( K_{sp} \). So that works. Wait, but why are there cube roots? Maybe a mistake in the problem, or maybe I misread the formula. But according to the given options, option a is \( 4.0 \times 10^{-6} \), which when squared gives \( 1.6 \times 10^{-11} \), so that's correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. \( 4.0 \times 10^{-6} \)