QUESTION IMAGE
Question
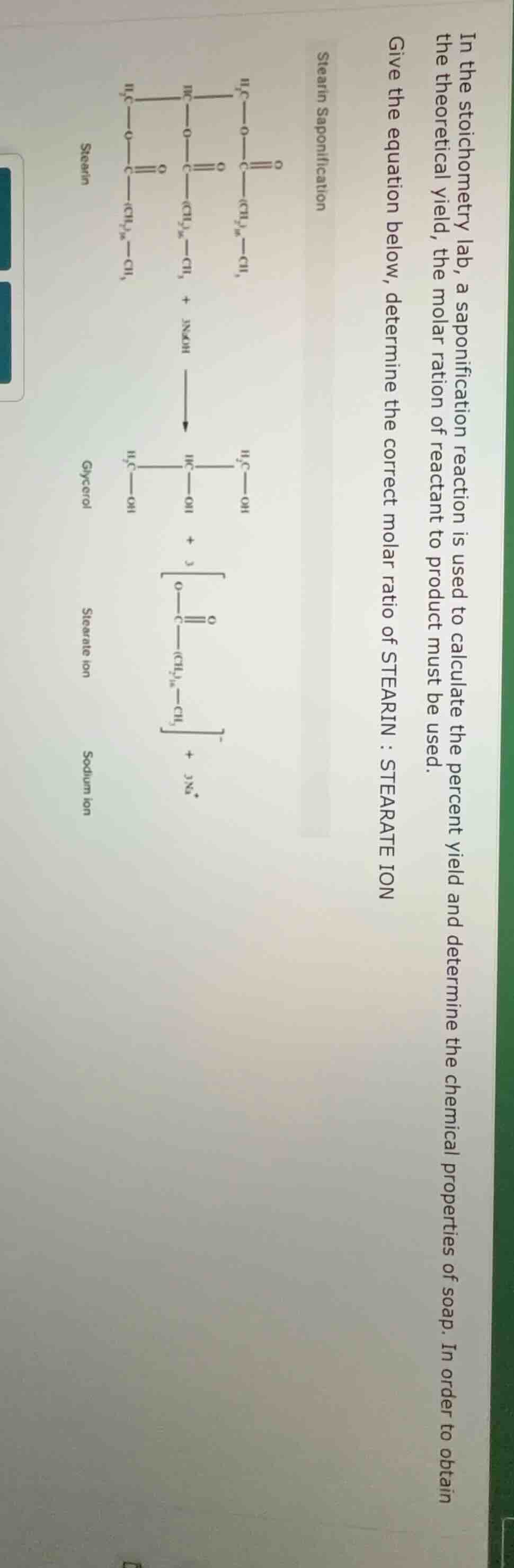
in the stoichometry lab, a saponification reaction is used to calculate the percent yield and determine the chemical properties of soap. in order to obtain the theoretical yield, the molar ration of reactant to product must be used. give the equation below, determine the correct molar ratio of stearin : stearate ion stearin saponification chemical structure of stearin reaction with naoh to form glycerol, stearate ion, and sodium ion, with corresponding chemical structures shown
Step1: Identify reactant and product moles
From the saponification equation, 1 mole of stearin reacts to produce 3 moles of stearate ion (looking at the coefficients: stearin has 1, stearate ion has 3 in the products).
Step2: Calculate molar ratio
Molar ratio of stearin to stearate ion is moles of stearin : moles of stearate ion = \(1 : 3\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(1:3\)