QUESTION IMAGE
Question
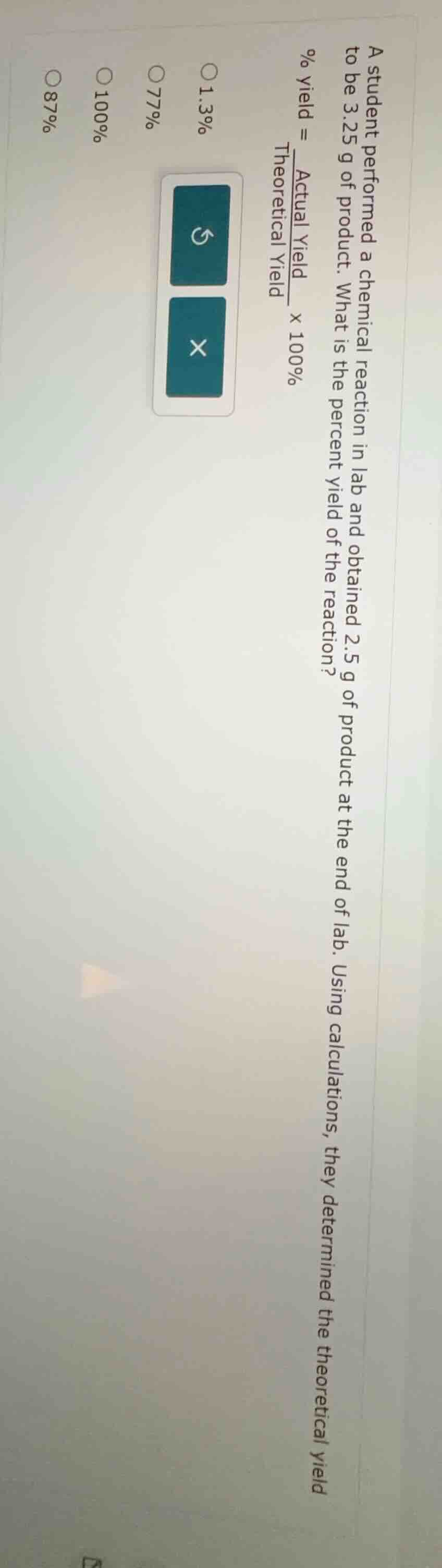
a student performed a chemical reaction in lab and obtained 2.5 g of product at the end of lab. using calculations, they determined the theoretical yield to be 3.25 g of product. what is the percent yield of the reaction?
% yield = \\(\frac{\text{actual yield}}{\text{theoretical yield}} \times 100\\%
\bigcirc 1.3\\%
\bigcirc 77\\%
\bigcirc 100\\%
\bigcirc 87\\%
Step1: Identify values
Actual Yield = 2.5 g, Theoretical Yield = 3.25 g
Step2: Apply % yield formula
% yield = $\frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100\%$ = $\frac{2.5}{3.25} \times 100\%$
Step3: Calculate the fraction
$\frac{2.5}{3.25} \approx 0.7692$
Step4: Multiply by 100%
$0.7692 \times 100\% \approx 77\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
77% (corresponding to the option with 77%)