QUESTION IMAGE
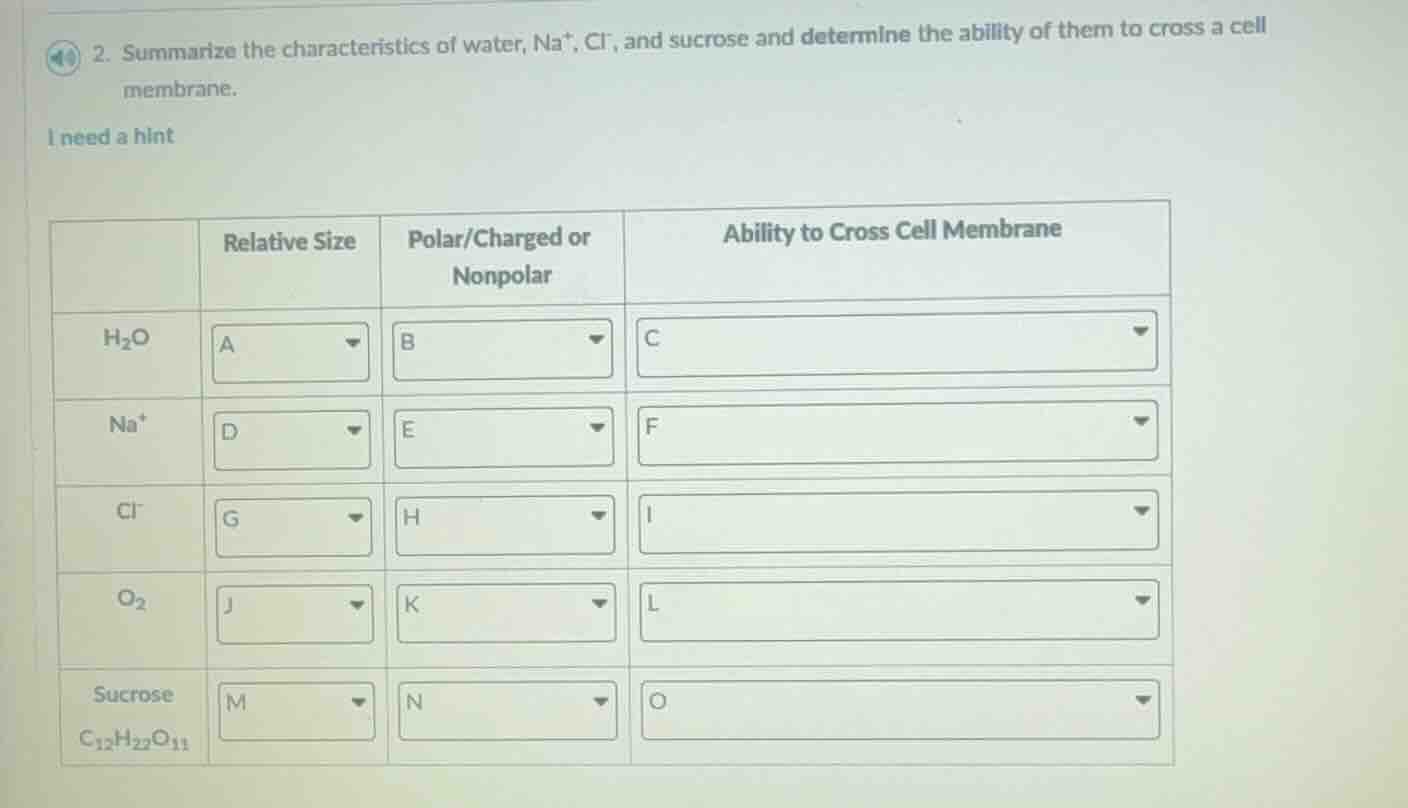
Question
- summarize the characteristics of water, na⁺, cl⁻, and sucrose and determine the ability of them to cross a cell membrane.
i need a hint
| relative size | polar/charged or nonpolar | ability to cross cell membrane | |
|---|---|---|---|
| na⁺ | d | e | f |
| cl⁻ | g | h | i |
| o₂ | j | k | l |
| sucrose c₁₂h₂₂o₁₁ | m | n | o |
Brief Explanations
Each substance's traits and membrane crossing ability are based on cell membrane permeability rules: small substances cross easier, charged/polar ones need help, nonpolar pass freely, and large polar molecules struggle without transporters.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Substance | Relative Size | Polar/Charged or Nonpolar | Ability to Cross Cell Membrane |
|---|---|---|---|
| $\text{Na}^+$ | Very small | Charged | Cannot cross passively; requires ion channels or active transport |
| $\text{Cl}^-$ | Very small | Charged | Cannot cross passively; requires ion channels or active transport |
| $\text{O}_2$ | Very small | Nonpolar | Freely diffuses across membrane |
| Sucrose ($\text{C}_{12}\text{H}_{22}\text{O}_{11}$) | Large | Polar | Cannot cross passively; requires facilitated diffusion or active transport |