QUESTION IMAGE
Question
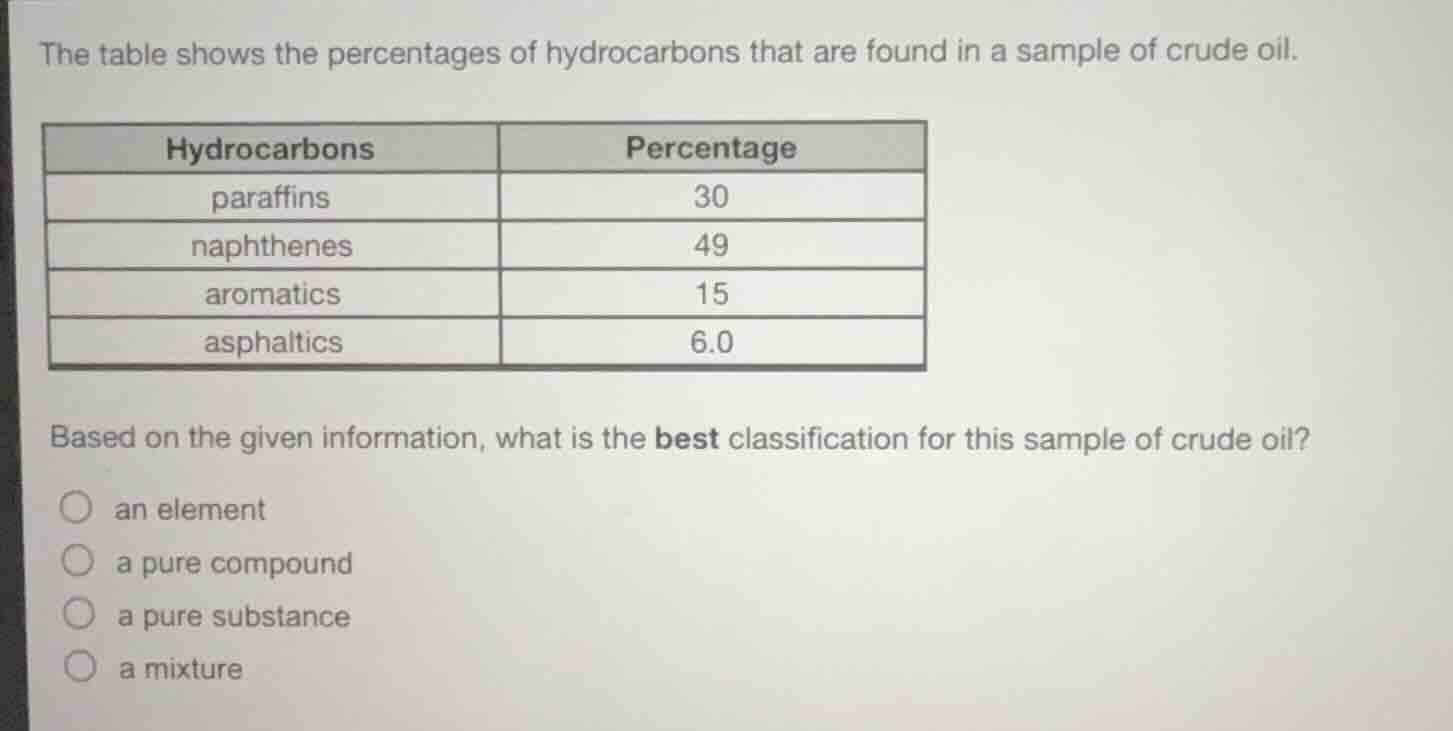
the table shows the percentages of hydrocarbons that are found in a sample of crude oil.
hydrocarbons | percentage
paraffins | 30
naphthenes | 49
aromatics | 15
asphalitics | 6.0
based on the given information, what is the best classification for this sample of crude oil?
○ an element
○ a pure compound
○ a pure substance
○ a mixture
Brief Explanations
- Recall the definitions: An element is a pure substance made of one type of atom. A pure compound is a substance with a fixed composition of two or more elements chemically bonded. A pure substance (element or compound) has a fixed composition. A mixture is a combination of two or more substances (elements or compounds) that are physically mixed, not chemically combined, and has variable composition.
- Analyze the crude oil sample: The table shows different hydrocarbons (paraffins, naphthenes, aromatics, asphaltic) with different percentages. This means the crude oil is made up of multiple substances (hydrocarbons) with varying proportions, which fits the definition of a mixture. It can't be an element (multiple substances), a pure compound (multiple substances with variable composition, not a fixed compound), or a pure substance (since pure substances have fixed composition, but here we have multiple components with different percentages).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. a mixture