QUESTION IMAGE
Question
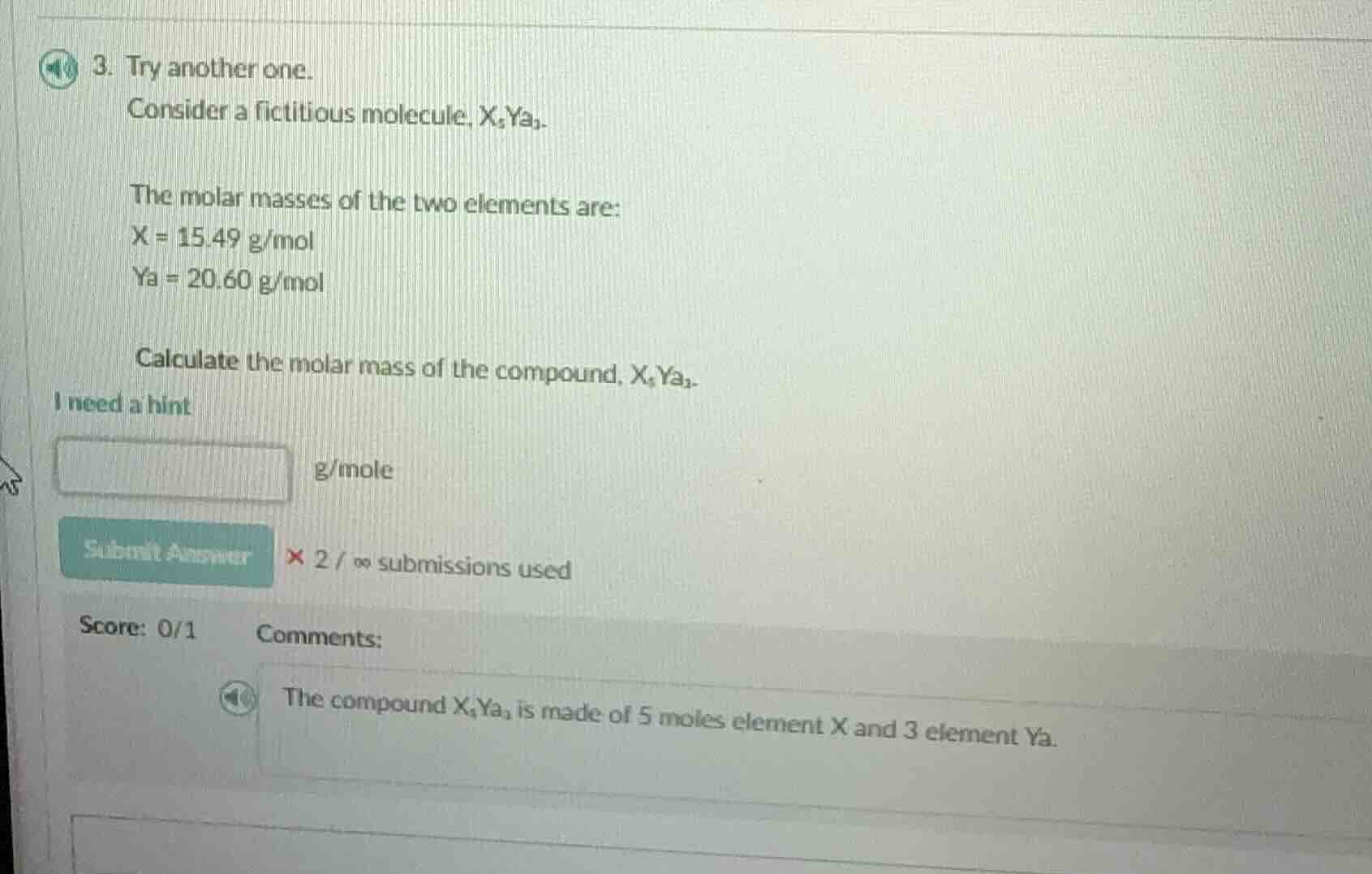
- try another one.
consider a fictitious molecule, x₅ya₃.
the molar masses of the two elements are:
x = 15.49 g/mol
ya = 20.60 g/mol
calculate the molar mass of the compound, x₅ya₃.
i need a hint
blank g/mole
submit answer × 2 / ∞ submissions used
score: 0/1 comments:
the compound x₅ya₃ is made of 5 moles element x and 3 element ya.
Step1: Calculate mass of X in compound
Molar mass of X is \(15.49\) g/mol, and there are 5 moles of X. So mass of X: \(5\times15.49 = 77.45\) g/mol.
Step2: Calculate mass of Ya in compound
Molar mass of Ya is \(20.60\) g/mol, and there are 3 moles of Ya. So mass of Ya: \(3\times20.60 = 61.80\) g/mol.
Step3: Sum masses to get molar mass of \(X_5Ya_3\)
Add mass of X and Ya: \(77.45 + 61.80 = 139.25\) g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(139.25\)