QUESTION IMAGE
Question
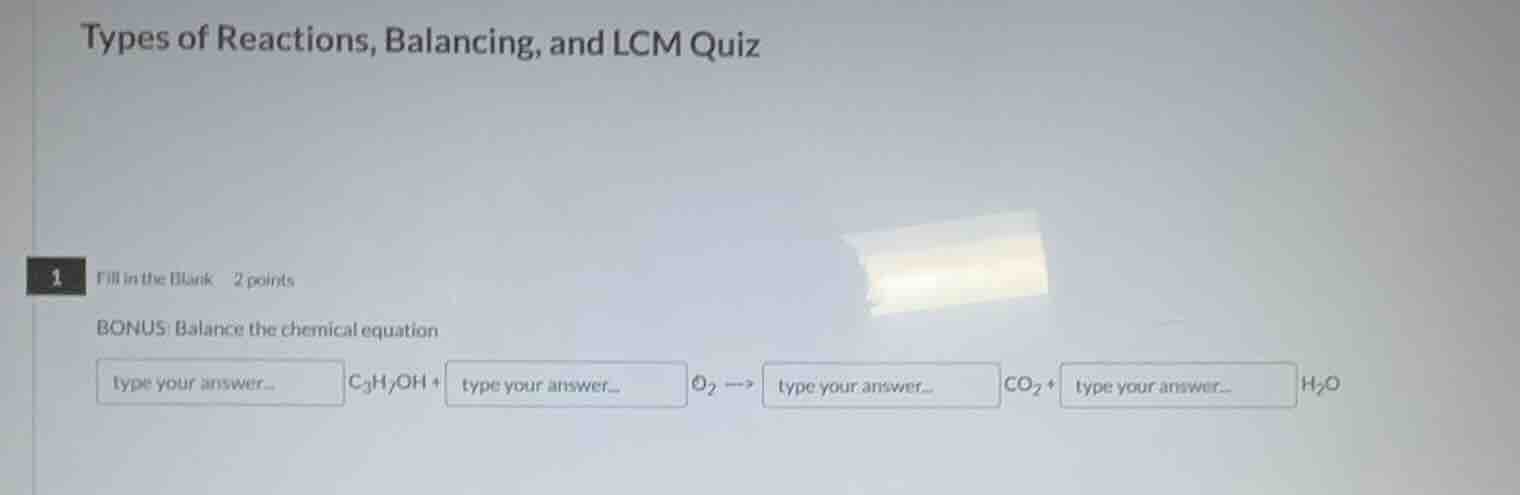
types of reactions, balancing, and lcm quiz
1 fill in the blank 2 points
bonus: balance the chemical equation
type your answer... c₃h₇oh + type your answer... o₂ → type your answer... co₂ + type your answer... h₂o
Step1: Count initial atoms
Reactants: $C_5H_7OH$ has 5 C, 8 H, 1 O; $O_2$ has 2 O. Products: $CO_2$ has 1 C, 2 O; $H_2O$ has 2 H, 1 O.
Step2: Balance C atoms
Assign coefficient 5 to $CO_2$ to match 5 C on left.
Step3: Balance H atoms
Assign coefficient 4 to $H_2O$ to match 8 H on left.
Step4: Balance O atoms
Calculate total O on right: $5\times2 + 4\times1 = 14$. Subtract 1 O from $C_5H_7OH$, so $O_2$ needs $\frac{13}{2}$.
Step5: Eliminate fraction
Multiply all coefficients by 2.
Step6: Verify atom counts
Reactants: 10 C, 16 H, 28 O. Products: 10 C, 16 H, 28 O. Balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2, 13, 10, 8