QUESTION IMAGE
Question
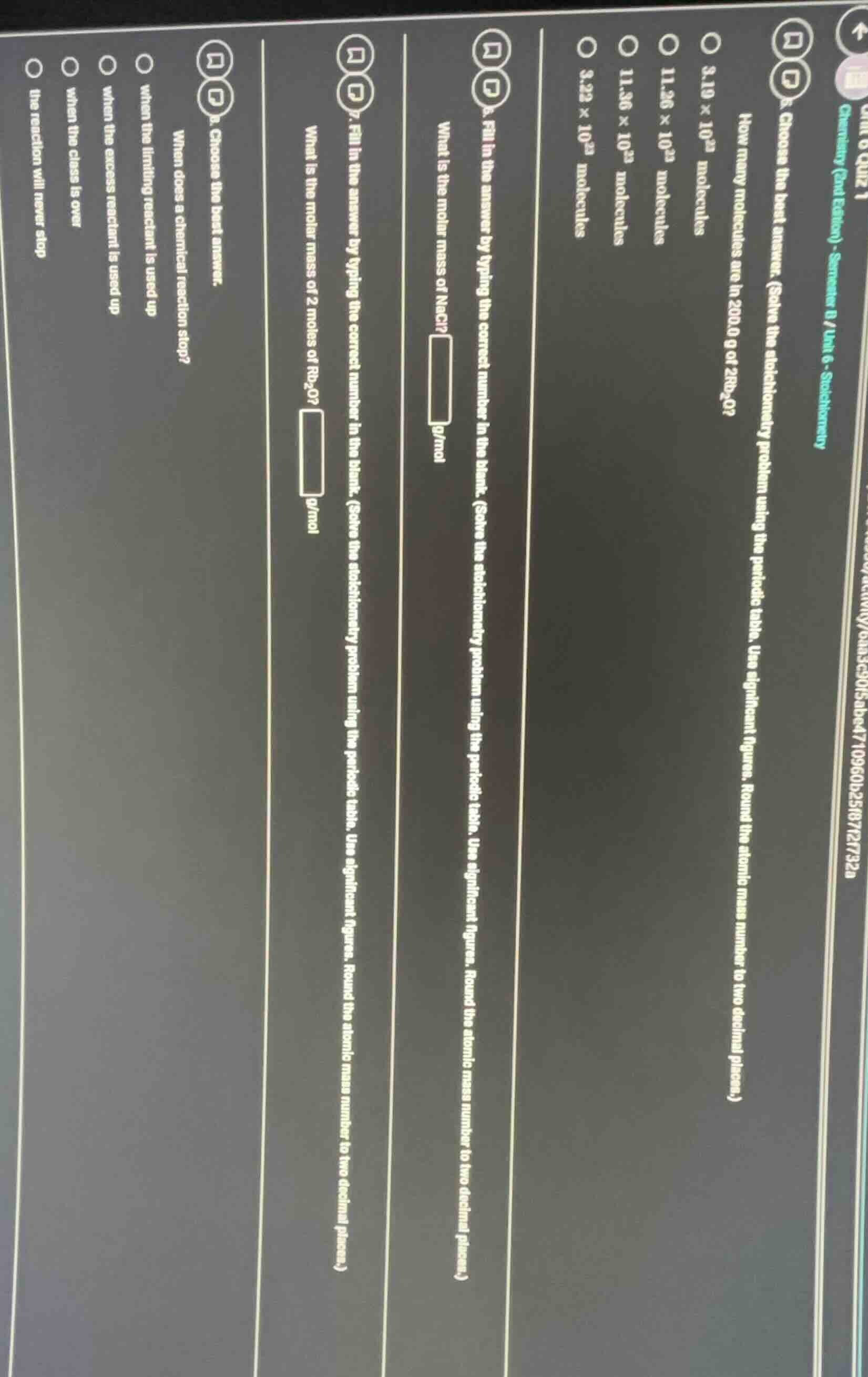
unit 6 quiz 1 chemistry (2nd edition) - semester b / unit 6 - stoichiometry choose the best answer. how many molecules are in 200.0 g of 2rb₂o? 3.10 × 10²³ molecules 11.26 × 10²³ molecules 11.36 × 10²³ molecules 3.23 × 10²³ molecules 6. fill in the answer by typing the correct number in the blank. (solve the stoichiometry problem using the periodic table. use significant figures. round the atomic mass number to two decimal places.) what is the molar mass of nacl? g/mol 7. fill in the answer by typing the correct number in the blank. (solve the stoichiometry problem using the periodic table. use significant figures. round the atomic mass number to two decimal places.) what is the molar mass of 2 moles of rb₂o? g/mol 8. choose the best answer. when does a chemical reaction stop? when the limiting reactant is used up when the excess reactant is used up when the class is over the reaction will never stop
Step1: Calculate molecules in 200.0 g Rb₂O
First, find molar mass of $\text{Rb}_2\text{O}$:
Molar mass Rb = 85.47 g/mol, O = 16.00 g/mol
Molar mass $\text{Rb}_2\text{O} = 2\times85.47 + 16.00 = 186.94$ g/mol
Moles of $\text{Rb}_2\text{O} = \frac{200.0}{186.94} \approx 1.070$ mol
Molecules = $1.070 \times 6.022\times10^{23} \approx 6.44\times10^{23}$? Correction: Recheck calculation
Wait, $200.0 / 186.94 = 1.070$, $1.070*6.022e23=6.44e23$? No, the options given:
Wait, recheck atomic mass: Rb is 85.4678, so 2*85.4678=170.9356 +16.00=186.9356
200/186.9356=1.070 mol, 1.070*6.02214e23=6.44e23, but none of the options match? Wait no, the options are 3.19, 11.26, 11.36, 3.22. Oh! Wait, I misread: 200.0 g of 2Rb₂O? No, the question is 200.0 g of Rb₂O. Wait, no, maybe I messed up:
Wait, $N = \frac{m}{M} \times N_A$
$M(\text{Rb}_2\text{O})=2\times85.47 +16.00=186.94$ g/mol
$N=\frac{200.0}{186.94} \times 6.022\times10^{23} \approx 1.070 \times 6.022\times10^{23} \approx 6.44\times10^{23}$. This is not in options. Wait, maybe the question is 200.0 g of Rb₂O, but maybe I used wrong atomic mass? Wait, if Rb is 87.62 (wrong, that's Sr), no. Wait, no, maybe the question is 200.0 g of RbO? No, the question says 2Rb₂O? No, the question says "200.0 g of 2Rb₂O" which is 2 moles of Rb₂O? No, 2 moles would be 2186.94=373.88 g. Wait, maybe the question is 200.0 g of Rb₂O, and the options are wrong? No, wait, I must have misread. Wait, the options are 3.19e23, 11.26e23, 11.36e23, 3.22e23. Oh! Wait, 200.0 g of Rb? No, the question says 2Rb₂O. Wait, no, let's check question again: "How many molecules are in 200.0 g of 2Rb₂O?" Oh! 2Rb₂O is 2 formula units, so molar mass of 2Rb₂O is 2186.94=373.88 g/mol. Then moles = 200.0 / 373.88 ≈0.535 mol. Molecules = 0.535 *6.022e23≈3.22e23. Oh! That's option D.
Step2: Molar mass of NaCl
Atomic mass Na=22.99 g/mol, Cl=35.45 g/mol
Molar mass $\text{NaCl}=22.99 + 35.45 = 58.44$ g/mol
Step3: Molar mass of 2 moles Rb₂O
Molar mass of 1 mole $\text{Rb}_2\text{O}=186.94$ g/mol
Molar mass of 2 moles $=2\times186.94=373.88$ g/mol
Step4: Reaction stop condition
A reaction stops when limiting reactant is consumed.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 3.22 × 10²³ molecules
- 58.44
- 373.88
- when the limiting reactant is used up