QUESTION IMAGE
Question
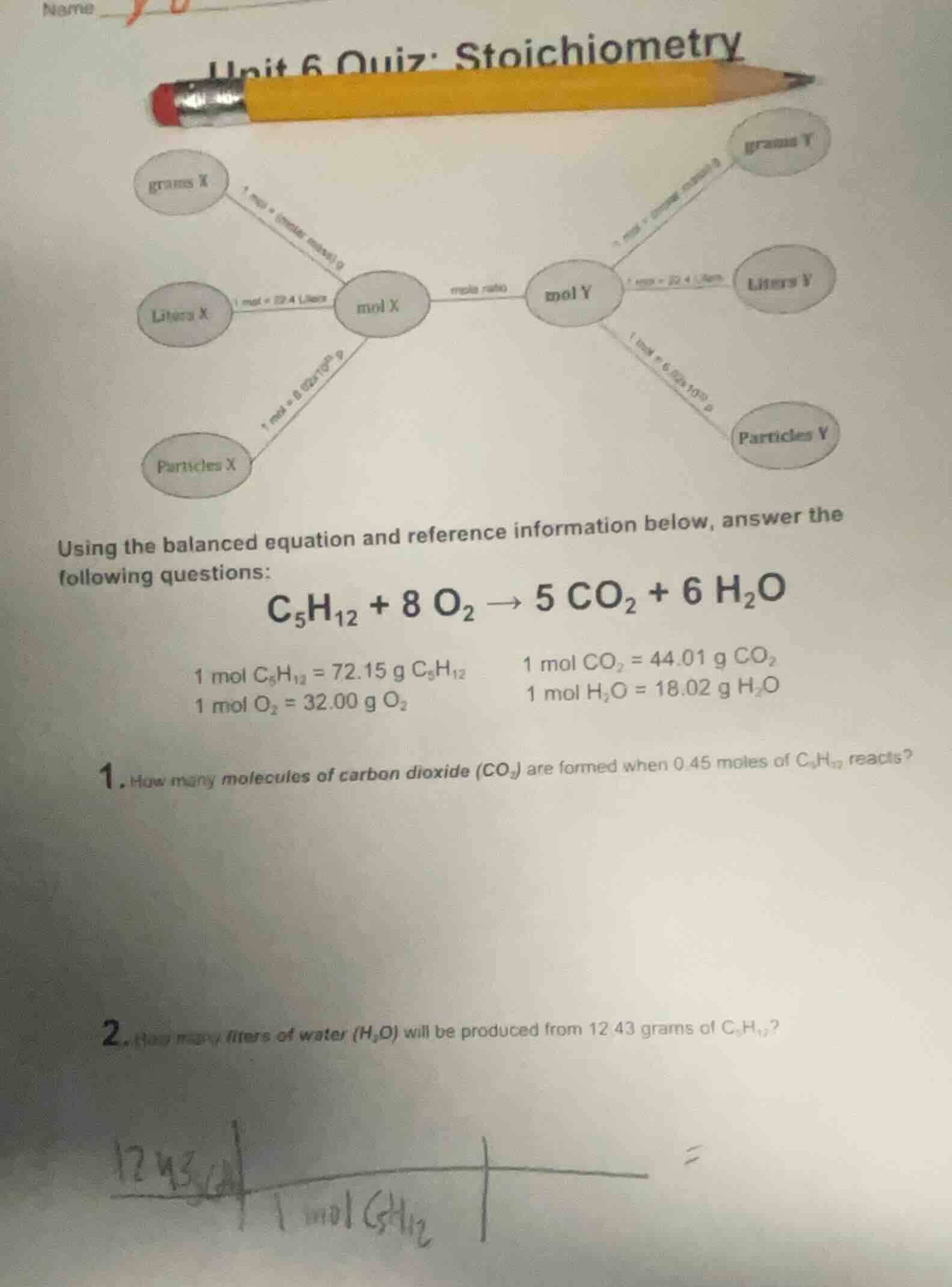
unit 6 quiz: stoichiometry
using the balanced equation and reference information below, answer the following questions:
$ce{c_{5}h_{12} + 8 o_{2} -> 5 co_{2} + 6 h_{2}o}$
1 mol $ce{c_{5}h_{12}}$ = 72.15 g $ce{c_{5}h_{12}}$ \t1 mol $ce{co_{2}}$ = 44.01 g $ce{co_{2}}$
1 mol $ce{o_{2}}$ = 32.00 g $ce{o_{2}}$ \t1 mol $ce{h_{2}o}$ = 18.02 g $ce{h_{2}o}$
- how many molecules of carbon dioxide ($ce{co_{2}}$) are formed when 0.45 moles of $ce{c_{5}h_{12}}$ reacts?
- how many liters of water ($ce{h_{2}o}$) will be produced from 12.43 grams of $ce{c_{5}h_{12}}$?
Step1: Find moles of $\text{CO}_2$
From the balanced equation, the mole ratio of $\text{C}_5\text{H}_{12}$ to $\text{CO}_2$ is $1:5$.
$\text{Moles of CO}_2 = 0.45\ \text{mol C}_5\text{H}_{12} \times \frac{5\ \text{mol CO}_2}{1\ \text{mol C}_5\text{H}_{12}} = 2.25\ \text{mol CO}_2$
Step2: Convert moles to molecules
Use Avogadro's number ($6.022 \times 10^{23}$ molecules/mol).
$\text{Molecules of CO}_2 = 2.25\ \text{mol} \times 6.022 \times 10^{23}\ \text{molecules/mol}$
Step3: Calculate final value
$\text{Molecules of CO}_2 = 1.355 \times 10^{24}$
---
Step1: Convert grams of $\text{C}_5\text{H}_{12}$ to moles
Use molar mass of $\text{C}_5\text{H}_{12}$ ($72.15\ \text{g/mol}$).
$\text{Moles of C}_5\text{H}_{12} = \frac{12.43\ \text{g}}{72.15\ \text{g/mol}} \approx 0.1723\ \text{mol}$
Step2: Find moles of $\text{H}_2\text{O}$
From balanced equation, mole ratio $\text{C}_5\text{H}_{12}:\text{H}_2\text{O} = 1:6$.
$\text{Moles of H}_2\text{O} = 0.1723\ \text{mol C}_5\text{H}_{12} \times \frac{6\ \text{mol H}_2\text{O}}{1\ \text{mol C}_5\text{H}_{12}} \approx 1.034\ \text{mol}$
Step3: Convert moles to liters
Assume STP: 1 mol gas = 22.4 L.
$\text{Liters of H}_2\text{O} = 1.034\ \text{mol} \times 22.4\ \text{L/mol}$
Step4: Calculate final value
$\text{Liters of H}_2\text{O} \approx 23.16\ \text{L}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $1.36 \times 10^{24}$ molecules of $\text{CO}_2$
- $23.2$ liters of $\text{H}_2\text{O}$