QUESTION IMAGE
Question
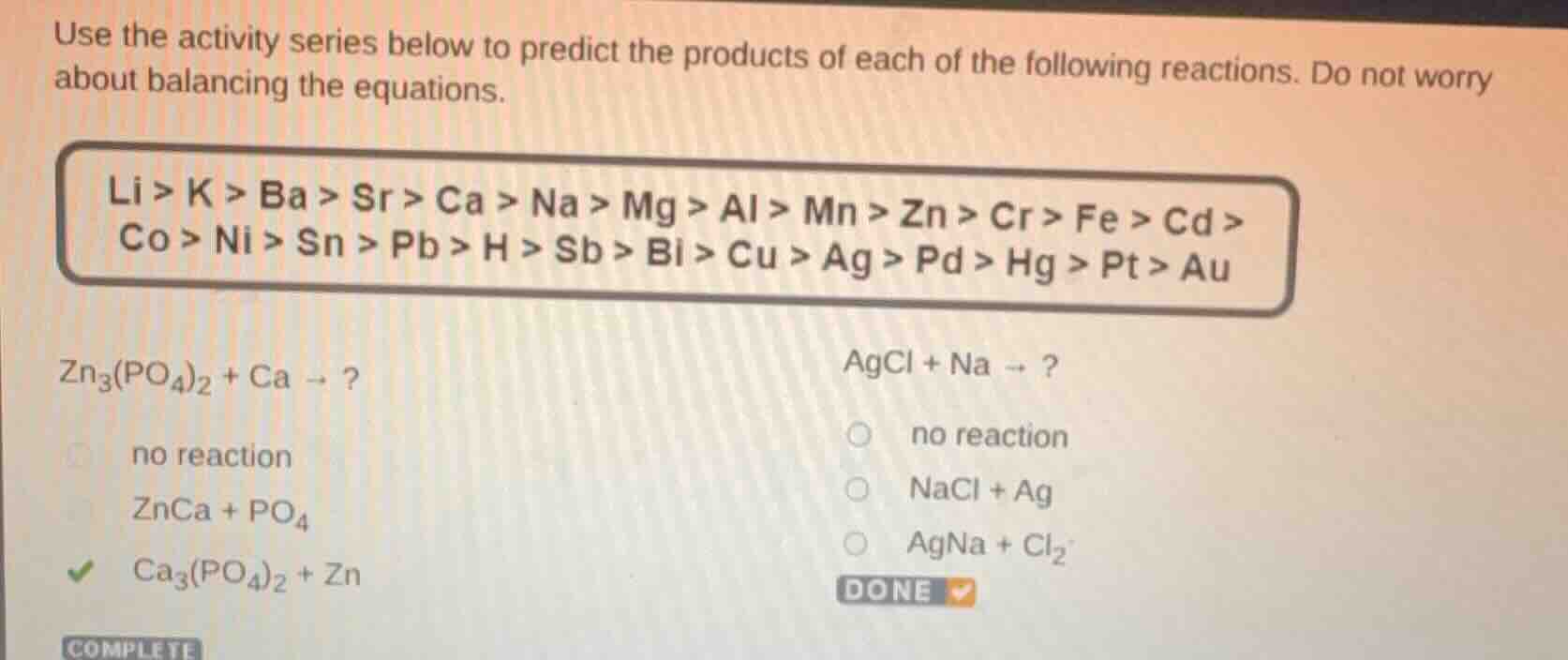
use the activity series below to predict the products of each of the following reactions. do not worry about balancing the equations.
li > k > ba > sr > ca > na > mg > al > mn > zn > cr > fe > cd > co > ni > sn > pb > h > sb > bi > cu > ag > pd > hg > pt > au
zn₃(po₄)₂ + ca → ?
no reaction
znca + po₄
ca₃(po₄)₂ + zn
agcl + na → ?
no reaction
nacl + ag
agna + cl₂
First Reaction ($\boldsymbol{Zn_3(PO_4)_2 + Ca
ightarrow}$):
Step1: Check Activity Series
From the activity series, $Ca > Zn$ (Calcium is more reactive than Zinc). In a single - displacement reaction, a more reactive metal can displace a less reactive metal from its compound. The general form of a single - displacement reaction for a metal - metal compound reaction is $M_1+M_2X
ightarrow M_1X + M_2$, where $M_1$ is more reactive than $M_2$.
Here, the reactants are $Zn_3(PO_4)_2$ (a compound of Zinc) and $Ca$ (Calcium). Since $Ca$ is more reactive than $Zn$, Calcium will displace Zinc from zinc phosphate.
Step2: Predict Products
The reaction will be $Zn_3(PO_4)_2+Ca
ightarrow Ca_3(PO_4)_2 + Zn$. The calcium phosphate ($Ca_3(PO_4)_2$) is formed because Calcium combines with the phosphate ion, and Zinc is displaced as a free metal.
Second Reaction ($\boldsymbol{AgCl + Na
ightarrow}$):
Step1: Check Activity Series
From the activity series, $Na > Ag$ (Sodium is more reactive than Silver). For the reaction between a metal and a metal - halide compound (in this case, $AgCl$ is a silver chloride, a compound of Silver), a more reactive metal (Sodium) can displace a less reactive metal (Silver) from its compound. The general form of the single - displacement reaction here is $M_1+M_2X
ightarrow M_1X+M_2$.
Step2: Predict Products
The reaction will be $AgCl + Na
ightarrow NaCl+Ag$. Sodium combines with the chloride ion to form sodium chloride ($NaCl$), and Silver is displaced as a free metal.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
For $Zn_3(PO_4)_2 + Ca$: $\boldsymbol{Ca_3(PO_4)_2 + Zn}$
For $AgCl + Na$: $\boldsymbol{NaCl + Ag}$