QUESTION IMAGE
Question
use your periodic table to fill in the chart and to answer the following questions.
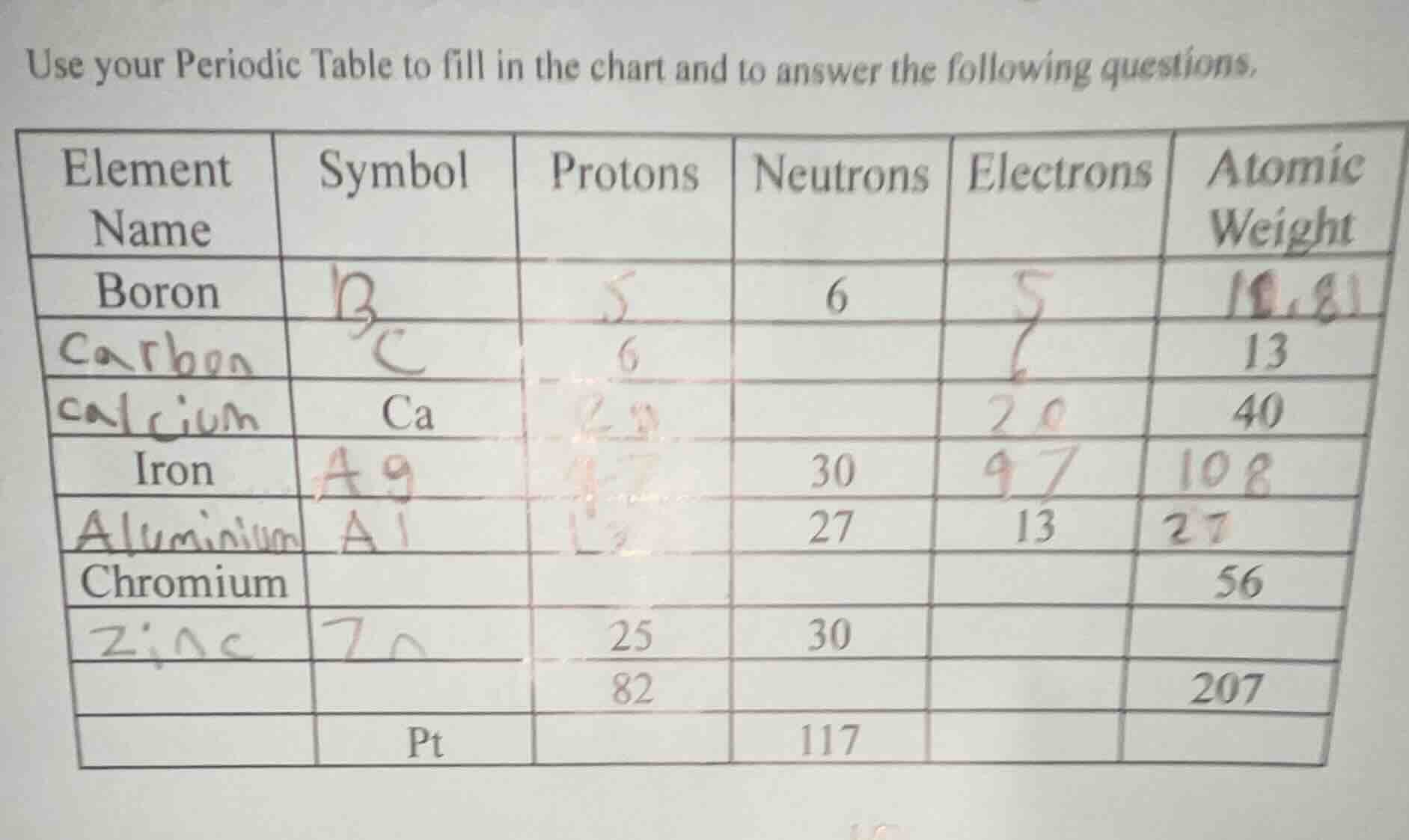
element name | symbol | protons | neutrons | electrons | atomic weight
boron | | | 6 | |
| c | 6 | 13 | ||
| ca | 40 |
iron | | | 30 | |
| al | 27 | 13 |
chromium | | | | | 56
| 25 | 30 | |||
| 82 | 207 | |||
| pt | 117 |
(note: some handwritten contents in the original chart are retained as they are, and the table structure is adjusted for clarity)
Step1: Recall key atomic rules
Protons = Atomic Number = Electrons (neutral atom); Atomic Weight ≈ Protons + Neutrons; Use periodic table for element data.
Step2: Fill Carbon row
Neutrons = Atomic Weight - Protons = $13 - 6 = 7$
Step3: Fill Calcium row
Protons = Atomic Number = 20; Neutrons = $40 - 20 = 20$
Step4: Correct Iron row
Symbol = Fe; Protons = 26; Neutrons = 30, so Atomic Weight = $26 + 30 = 56$; Electrons = 26
Step5: Fill Aluminium row
Protons = 13; Atomic Weight = $13 + 27 = 40$
Step6: Fill Chromium row
Symbol = Cr; Protons = 24; Electrons = 24; Neutrons = $56 - 24 = 32$
Step7: Fix Zinc row
Element Name = Manganese; Symbol = Mn; Electrons = 25; Atomic Weight = $25 + 30 = 55$
Step8: Fill 82-proton row
Element Name = Lead; Symbol = Pb; Electrons = 82; Neutrons = $207 - 82 = 125$
Step9: Fill Platinum row
Element Name = Platinum; Protons = 78; Electrons = 78; Atomic Weight = $78 + 117 = 195$
Step10: Correct Boron row
Atomic Weight = $5 + 6 = 11$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element Name | Symbol | Protons | Neutrons | Electrons | Atomic Weight |
|---|---|---|---|---|---|
| Carbon | C | 6 | 7 | 6 | 13 |
| Calcium | Ca | 20 | 20 | 20 | 40 |
| Iron | Fe | 26 | 30 | 26 | 56 |
| Aluminium | Al | 13 | 27 | 13 | 40 |
| Chromium | Cr | 24 | 32 | 24 | 56 |
| Manganese | Mn | 25 | 30 | 25 | 55 |
| Lead | Pb | 82 | 125 | 82 | 207 |
| Platinum | Pt | 78 | 117 | 78 | 195 |