QUESTION IMAGE
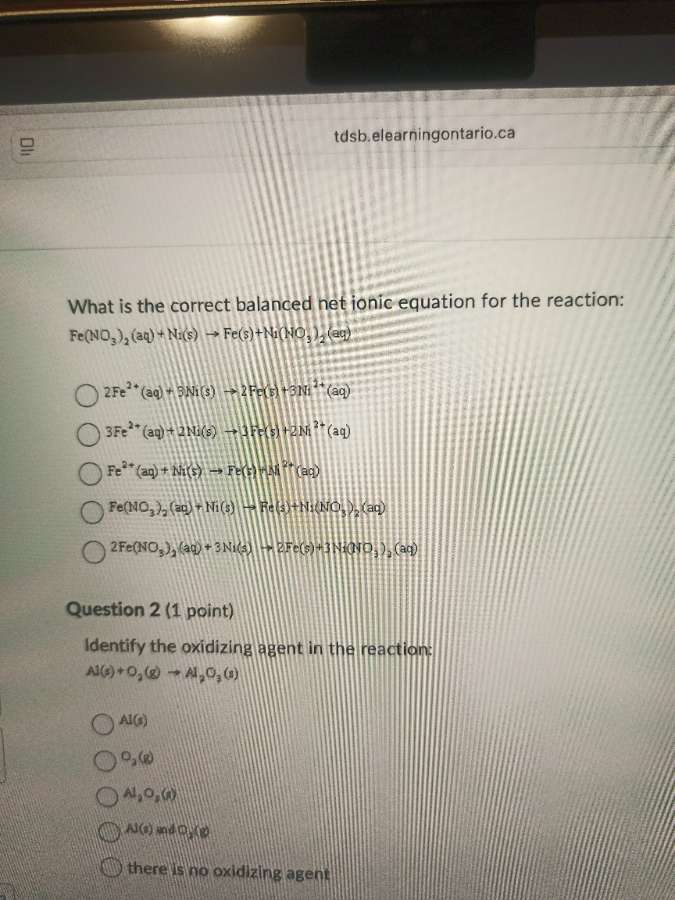
Question
what is the correct balanced net ionic equation for the reaction:
fe(no₃)₂(aq) + ni(s) → fe(s) + ni(no₃)₂(aq)
2fe²⁺(aq) + 3ni(s) → 2fe(s) + 3ni²⁺(aq)
3fe²⁺(aq) + 2ni(s) → 3fe(s) + 2ni²⁺(aq)
fe²⁺(aq) + ni(s) → fe(s) + ni²⁺(aq)
fe(no₃)₂(aq) + ni(s) → fe(s) + ni(no₃)₂(aq)
2fe(no₃)₂(aq) + 3ni(s) → 2fe(s) + 3ni(no₃)₂(aq)
question 2 (1 point)
identify the oxidizing agent in the reaction:
al(s) + o₂(g) → al₂o₃(s)
al(s)
o₂(g)
al₂o₃(s)
al(s) and o₂(g)
there is no oxidizing agent
Question 1: Balanced Net Ionic Equation
Step 1: Write the ionic form of reactants and products
The reactants are $\ce{Fe(NO_{3})_{2}(aq)}$ which dissociates into $\ce{Fe^{2+}(aq)}$ and $\ce{2NO_{3}^{-}(aq)}$, and $\ce{Ni(s)}$ (solid, so no dissociation). The products are $\ce{Fe(s)}$ (solid) and $\ce{Ni(NO_{3})_{2}(aq)}$ which dissociates into $\ce{Ni^{2+}(aq)}$ and $\ce{2NO_{3}^{-}(aq)}$.
Step 2: Cancel spectator ions (NO₃⁻)
The nitrate ions ($\ce{NO_{3}^{-}}$) are spectator ions as they appear on both sides. So we remove them.
Step 3: Balance the equation
The unbalanced ionic equation is $\ce{Fe^{2+}(aq) + Ni(s) -> Fe(s) + Ni^{2+}(aq)}$. Wait, no, wait the original reaction is $\ce{Fe(NO_{3})_{2}(aq) + Ni(s) -> Fe(s) + Ni(NO_{3})_{2}(aq)}$? Wait, no, the original reaction given is $\ce{Fe(NO_{3})_{2}(aq) + Ni(s) -> Fe(s) + Ni(NO_{3})_{2}(aq)}$? Wait, no, the user's first reaction is $\ce{Fe(NO_{3})_{2}(aq) + Ni(s) -> Fe(s) + Ni(NO_{3})_{2}(aq)}$? Wait, no, looking at the options, let's re-express. Wait, the first reaction: $\ce{Fe(NO_{3})_{2}(aq) + Ni(s) -> Fe(s) + Ni(NO_{3})_{2}(aq)}$? Wait, no, the options have different coefficients. Wait, maybe the original reaction was $\ce{2Fe(NO_{3})_{3}(aq) + 3Ni(s) -> 2Fe(s) + 3Ni(NO_{3})_{2}(aq)}$? No, the options: let's check the charges. Wait, $\ce{Fe(NO_{3})_{2}}$ has $\ce{Fe^{2+}}$, $\ce{Ni(NO_{3})_{2}}$ has $\ce{Ni^{2+}}$. Wait, no, the first option: $2\ce{Fe^{2+}(aq)} + 3\ce{Ni(s)} -> 2\ce{Fe(s)} + 3\ce{Ni^{2+}(aq)}$? Wait, no, the correct approach: first, write the molecular equation, then ionic, then net ionic.
Wait, the given reaction: $\ce{Fe(NO_{3})_{2}(aq) + Ni(s) -> Fe(s) + Ni(NO_{3})_{2}(aq)}$? But that would be a 1:1 ratio. But the options have different coefficients. Wait, maybe a typo, and the reactant is $\ce{Fe(NO_{3})_{3}}$? No, the options have $\ce{Fe^{2+}}$. Wait, let's check the options. The third option is $\ce{Fe^{2+}(aq) + Ni(s) -> Fe(s) + Ni^{2+}(aq)}$. But that's unbalanced? Wait, no, the charges: $\ce{Fe^{2+}}$ (oxidation state +2) becomes $\ce{Fe(s)}$ (0), so it's reduced. $\ce{Ni(s)}$ (0) becomes $\ce{Ni^{2+}}$ (+2), oxidized. The number of atoms: 1 Fe on left, 1 on right; 1 Ni on left, 1 on right. Charge: left side: +2 (from $\ce{Fe^{2+}}$) + 0 (Ni) = +2; right side: 0 (Fe) + +2 (Ni²⁺) = +2. So it's balanced. But wait, the original reaction: $\ce{Fe(NO_{3})_{2}(aq) + Ni(s) -> Fe(s) + Ni(NO_{3})_{2}(aq)}$. The ionic equation: $\ce{Fe^{2+}(aq) + 2NO_{3}^{-}(aq) + Ni(s) -> Fe(s) + Ni^{2+}(aq) + 2NO_{3}^{-}(aq)}$. Cancel $\ce{NO_{3}^{-}}$, so net ionic: $\ce{Fe^{2+}(aq) + Ni(s) -> Fe(s) + Ni^{2+}(aq)}$, which is the third option. Wait, but maybe the original reaction was $\ce{2Fe(NO_{3})_{2}(aq) + 3Ni(s) -> 2Fe(s) + 3Ni(NO_{3})_{2}(aq)}$? No, the options: let's check the first option: $2\ce{Fe^{2+}} + 3\ce{Ni} -> 2\ce{Fe} + 3\ce{Ni^{2+}}$. Charge: left: 2(+2) + 0 = +4; right: 0 + 3(+2) = +6. Not balanced. Second option: $3\ce{Fe^{2+}} + 2\ce{Ni} -> 2\ce{Fe} + 2\ce{Ni^{2+}}$. Charge: 3(+2)=+6, 0; right: 0 + 2(+2)=+4. Not balanced. Third option: $\ce{Fe^{2+}} + \ce{Ni} -> \ce{Fe} + \ce{Ni^{2+}}$. Charge: +2 + 0 = 0 + +2. Balanced. Atoms: 1 Fe, 1 Ni on both sides. So the third option is correct. Wait, but the original reaction's formula: $\ce{Fe(NO_{3})_{2}}$ and $\ce{Ni(NO_{3})_{2}}$, so the net ionic is $\ce{Fe^{2+}(aq) + Ni(s) -> Fe(s) + Ni^{2+}(aq)}$.
Step 4: Eliminate incorrect options
- Option 1: $2\ce{Fe^{2+}} + 3\ce{Ni} -> 2\ce{Fe} + 3\ce{Ni^{2+}}$: Charge not balanced (left +4, right +6).
- Option 2: $3\ce{Fe^{2+}} + 2\ce{Ni} -> 2\ce{Fe} + 2\ce{Ni^{2+}}$: Charge not balance…
Step 1: Recall the definition of oxidizing agent
An oxidizing agent is a substance that gets reduced (gains electrons, oxidation state decreases) and causes another substance to be oxidized (loses electrons, oxidation state increases).
Step 2: Determine oxidation states
- In $\ce{Al(s)}$, oxidation state of Al is 0.
- In $\ce{O_{2}(g)}$, oxidation state of O is 0.
- In $\ce{Al_{2}O_{3}(s)}$, oxidation state of Al: let x be oxidation state of Al. 2x + 3*(-2) = 0 → 2x -6 = 0 → x = +3. Oxidation state of O is -2.
Step 3: Identify reduction (oxidizing agent)
- Al: 0 → +3 (oxidized, loses electrons)
- O: 0 → -2 (reduced, gains electrons)
So $\ce{O_{2}(g)}$ is reduced, so it is the oxidizing agent.
Step 4: Eliminate incorrect options
- $\ce{Al(s)}$: oxidized, so reducing agent.
- $\ce{Al_{2}O_{3}(s)}$: product, not the agent.
- $\ce{Al(s)}$ and $\ce{O_{2}(g)}$: only $\ce{O_{2}}$ is oxidizing agent.
- "there is no oxidizing agent": incorrect, since redox reaction occurs.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Question 1): $\boldsymbol{\ce{Fe^{2+}(aq) + Ni(s) -> Fe(s) + Ni^{2+}(aq)}}$ (the third option)