QUESTION IMAGE
Question
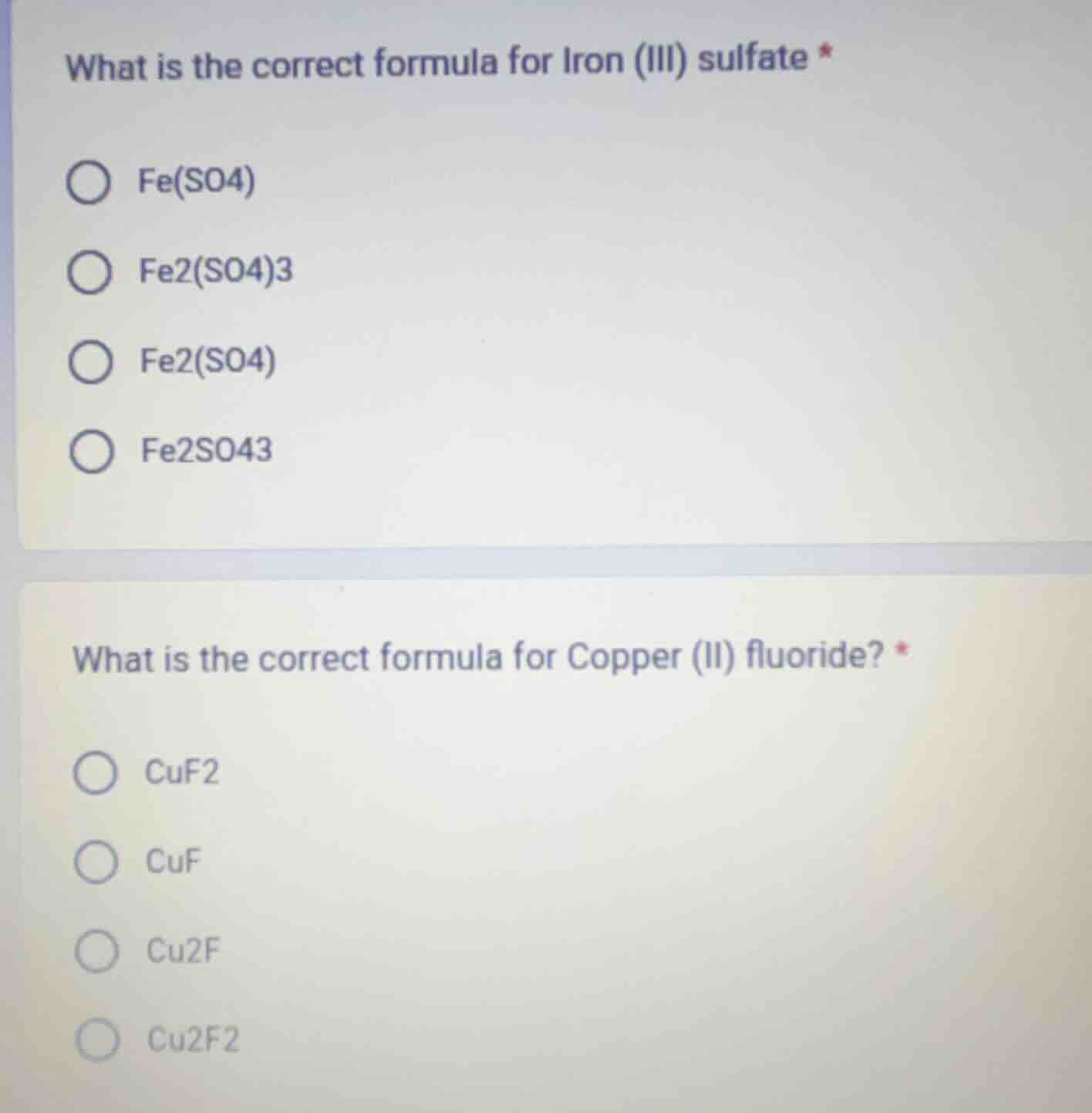
what is the correct formula for iron (iii) sulfate *
fe(so4)
fe2(so4)3
fe2(so4)
fe2so43
what is the correct formula for copper (ii) fluoride? *
cuf2
cuf
cu2f
cu2f2
First Question: What is the correct formula for Iron (III) sulfate?
To determine the formula for Iron (III) sulfate, we first identify the ions. Iron (III) means $\text{Fe}^{3+}$, and the sulfate ion is $\text{SO}_4^{2-}$. To balance the charges, we use the criss - cross method. The charge of Fe ($+3$) becomes the subscript of $\text{SO}_4$, and the charge of $\text{SO}_4$ ($-2$) becomes the subscript of Fe. So we get $\text{Fe}_2(\text{SO}_4)_3$ (since $3\times2 = 2\times3$ to balance the positive and negative charges: $2\times(+3)+3\times(-2)=6 - 6 = 0$).
Copper (II) is $\text{Cu}^{2+}$ and the fluoride ion is $\text{F}^-$. Using the criss - cross method, the charge of Cu ($+2$) becomes the subscript of F, and the charge of F ($-1$) becomes the subscript of Cu. So we get $\text{CuF}_2$ (because $1\times(+2)+2\times(-1)=2 - 2 = 0$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{Fe}_2(\text{SO}_4)_3$