QUESTION IMAGE
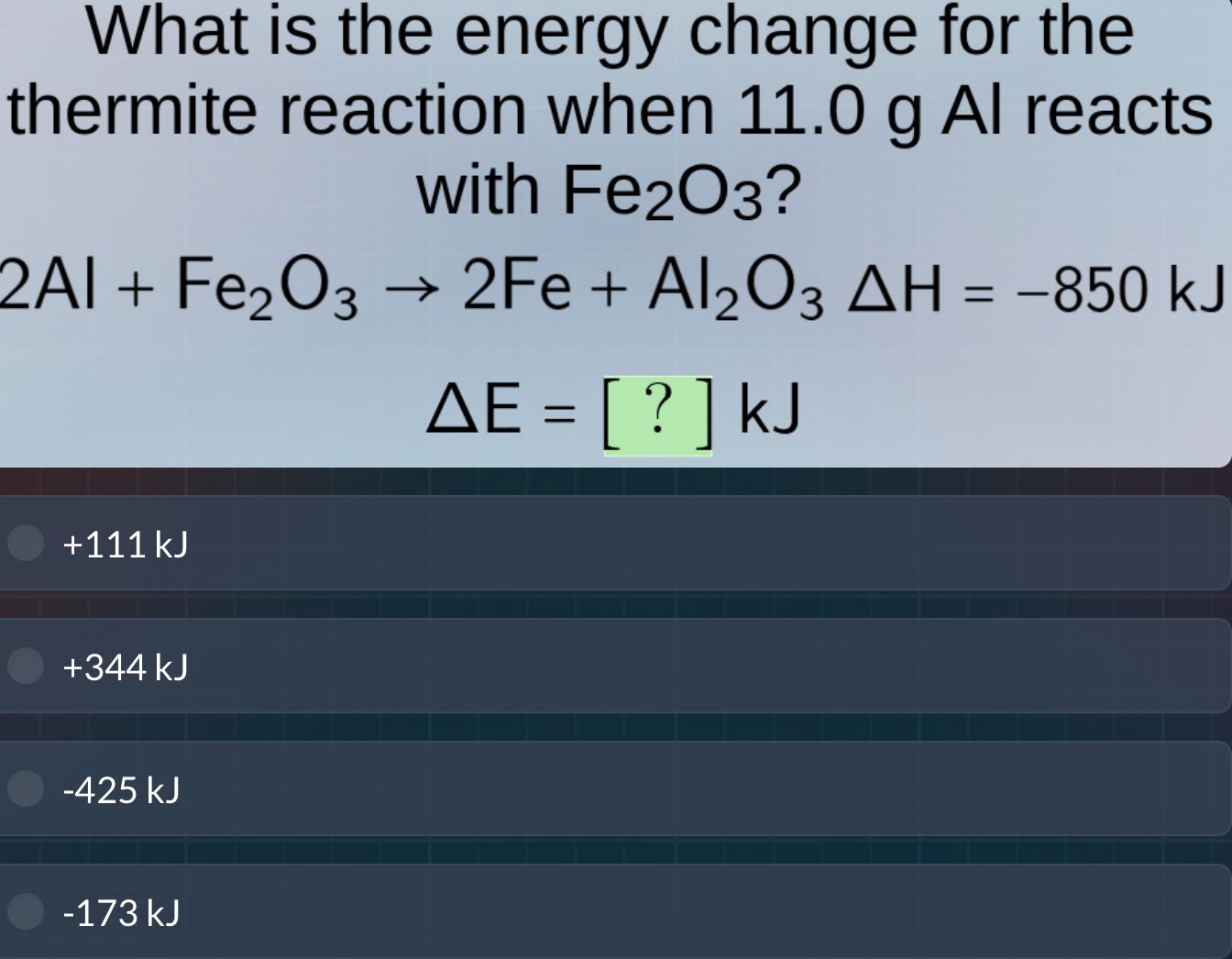
Question
what is the energy change for the thermite reaction when 11.0 g al reacts with fe₂o₃?
2al + fe₂o₃ → 2fe + al₂o₃ δh = -850 kj
δe = ? kj
+111 kj
+344 kj
-425 kj
-173 kj
Step1: Calculate moles of Al
Molar mass of Al is 27.0 g/mol. Moles of Al = $\frac{11.0\ \text{g}}{27.0\ \text{g/mol}} \approx 0.407\ \text{mol}$.
Step2: Relate moles to reaction enthalpy
From the reaction, 2 moles of Al release 850 kJ (since $\Delta H = -850\ \text{kJ}$, exothermic). For 0.407 mol of Al, energy change $\Delta E$ (approx equal to $\Delta H$ for reactions with small volume change) is calculated as: $\Delta E = \frac{0.407\ \text{mol}}{2\ \text{mol}} \times (-850\ \text{kJ}) \approx -173\ \text{kJ}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
-173 kJ (corresponding to the option: -173 kJ)