QUESTION IMAGE
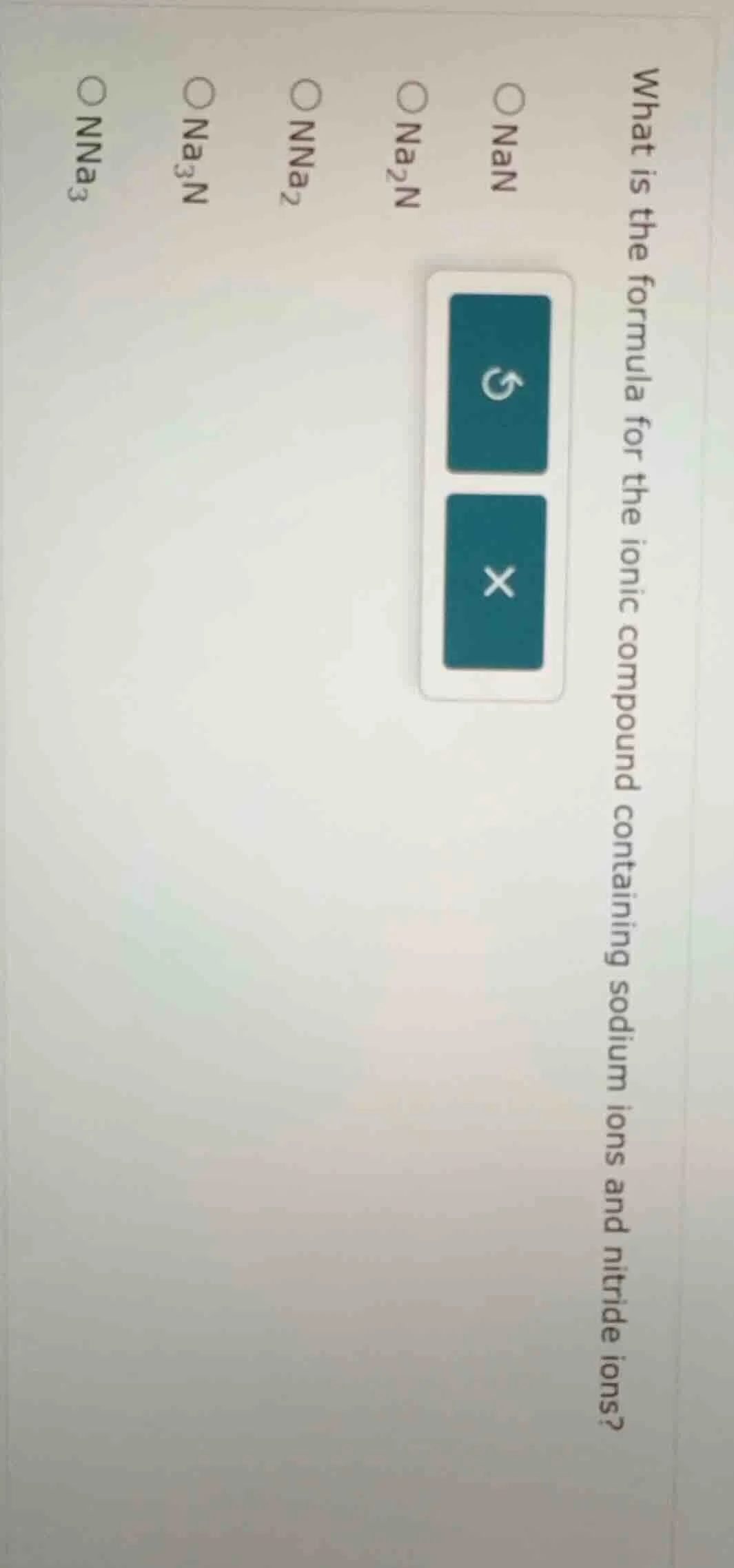
Question
what is the formula for the ionic compound containing sodium ions and nitride ions?
○ nan
○ na₂n
○ nna₂
○ na₃n
○ nna₃
Step1: Determine ion charges
Sodium ion is \( \text{Na}^+ \) (charge +1), nitride ion is \( \text{N}^{3-} \) (charge -3).
Step2: Balance charges
To balance positive and negative charges, we need 3 \( \text{Na}^+ \) ions (total +3) to balance 1 \( \text{N}^{3-} \) ion (total -3).
Step3: Write formula
The formula is \( \text{Na}_3\text{N} \) (matching the option "Na₃N").
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Na₃N (the option labeled "Na₃N")